
Svensk Kärnbränslehantering AB
Swedish Nuclear Fuel
and Waste Management Co
Box 250, SE-101 24 Stockholm
Phone +46 8 459 84 00
Technical Report
TR-10-69
Critical review of the literature on
the corrosion of copper by water
Fraser King
Integrity Corrosion Consulting Limited
December 2010
CM Gruppen AB, Bromma, 2011

Tänd ett lager:
P, R eller TR.
Critical review of the literature on
the corrosion of copper by water
Fraser King
Integrity Corrosion Consulting Limited
December 2010
ISSN 1404-0344
SKB TR-10-69
Keywords: Copper, Water, Hydrogen Evolution, Canisters, Lifetime Prediction.
This report concerns a study which was conducted for SKB. The conclusions
and viewpoints presented in the report are those of the author. SKB may draw
modied conclusions, based on additional literature sources and/or expert opinions.
A pdf version of this document can be downloaded from www.skb.se.
2 TR-10-69
The following figures are reproduced with permission of the copyright holder:
Figures 1-1, 2-7 and 2-8 with permission of NACE International, Houston, TX, USA.
Figures 1-2 and 3-5 with permission of the Nuclear Waste Management Organisation, Toronto, Canada.
Figures 2-2, 3-1, 3-2, 3-3, 3-4 and 3-6 with permission of Elsevier B.V., Amsterdam, The Netherlands.
Figure 2-3 with permission of Trans Tech Publications, Zurich, Switzerland.
Figures 2-4, 2-5 and 2-6 with permission of The Electrochemical Society, Pennington, NJ, USA.
Figures 2-9 and 2-10 with permission of Springer, Dordrecht, The Netherlands.
TR-10-69 3
Abstract
The conventional belief that copper is thermodynamically stable in oxygen-free water has been
questioned by a research group from the Royal Institute of Technology, Stockholm lead by
Dr. Gunnar Hultquist.
A critical review of the literature both in support of the proposed mechanism and that which argues
against it has been conducted. The critical review has been supported by supplementary analyses,
with particular focus on the scientific validity of the reported observations and their significance for
the corrosion of a copper canister.
It is found that:
• thescientificevidenceinsupportofthesuggestionthatwateroxidisescopperisnotconclusive
and there are many aspects which are unclear and contradictory,
• despiteanumberofattempts,nootherresearchershavebeenabletoreproducetheobservations
of Hultquist and co-workers,
• evenifcorrect,themechanismisnotimportantforcoppercanistersinarepository,bothbecause
of differences in the environmental conditions and because, even if corrosion did occur by this
mechanism, it would quickly stop, and
• thereisnoadverseimpactonthelifetimeofcoppercanistersduetothisproposed,butunproven,
mechanism.
TR-10-69 5
Contents
1 Introduction 7
2 Critical review of the literature 11
2.1 Critical review 11
2.1.1 The original article of Hultquist, Corrosion Science 1986 11
2.1.2 Simpson and Schenk, Corrosion Science 1987 14
2.1.3 Seo et al. Proc. 10
th
ICC, 1987 14
2.1.4 Eriksen et al. SKB TR-88-17 and Corrosion Science 1989 16
2.1.5 Hultquist et al. Corrosion Science 1989 16
2.1.6 Möller, SKI Report 95:72 19
2.1.7 Gråsjö et al. Mater. Sci. Forum 1995 20
2.1.8 Szakálos et al. Electrochem. Solid-State Letters 2007 20
2.1.9 Johansson, Electrochem. Solid-State Letters 2008 23
2.1.10 Hultquist et al. 17
th
Int. Corrosion Congress 2008 23
2.1.11 Report from the SSM BRITE group 2009 25
2.1.12 Hultquist et al. Catal. Lett. 2009 27
2.1.13 Korzhavyi and Johansson, SKB TR-10-30, 2010 30
2.1.14 Bojinov et al. Corros. Sci. 2010 30
2.2 Summary of key points from the critical review of the literature 31
3 Supplementary analyses 33
3.1 Formation and properties of adsorbed Cu-OH species 33
3.2 Corrosion of copper in anoxic concentrated chloride solutions 38
3.3 Corrosion of copper in compacted bentonite 38
3.4 Role of dissolved oxygen 39
3.5 Corrosion potential considerations 41
3.6 Implications for corrosion of a copper canister in a deep geologic repository 42
4 Implications for canister lifetimes 43
5 Summary and conclusions 47
References 49
TR-10-69 7
1 Introduction
The integrity of the copper-cast iron canister is an important aspect of the KBS-3 repository design.
For the expected repository conditions, analyses based on nearly three decades of research indicate
that a 5-cm-thick copper shell will not fail within 100,000 years (SKB 2006a, b). Corrosion will
be minimal because of the limited amount of oxidant within the repository and because of mass-
transport limitations imposed by the use of highly compacted bentonite around the canisters. Once
the initially trapped O
2
in the bentonite (and any Cu(II) produced by the homogeneous oxidation of
Cu(I) to Cu(II)) has been consumed, corrosion of the canister will cease unless sulphide reaches the
canister surface (King et al. 2001, SKB 2006a, b). Based on the amount of sulphide present in the
deposition holes and the amount that can diffuse to the canister surface from the groundwater, the
estimated depth of corrosion after 100,000 yrs is <5 mm (SKB 2006a, b).
Long-term containment of the spent fuel is achieved through a series of engineered barriers, of
which the copper canister is an integral component. These barriers, including the canister and
clay-based buffer and backfill sealing materials, are designed to perform as an integrated system,
one function of which is to ensure long canister lifetimes. A number of factors contribute to the long
canister lifetimes, including:
• thelimitedrateofmasstransportofreactantsto,andofcorrosionproductsawayfrom,the
canister surface in highly compacted bentonite (HCB),
• thesuppressionofmicrobialactivitybyHCB,
• thelimitedavailabilityofoxidantwithintherepositoryenvironment,
• theinsignificantradiationfieldandyieldofradiolysisproductsresultingfromtheuseofathick-
walled canister,
• thethermodynamicstabilityofcopperinO
2
-free aqueous solutions (in the absence of sulphide),
• thelimitedavailabilityofsulphideintheHCBandgroundwater,
• theabsenceofrapidformsoflocalisedcorrosionandenvironmentallyassistedcrackinginthe
expected repository environment, and
• thestructuralstabilityofthecanisterresultingfromtheuseofathick-walledcastironinsert.
Thus, the thermodynamic stability of copper in O
2
-free aqueous environments is only one of a
number of factors that lead to the long predicted canister lifetimes. Even then, the possibility that
H
2
O (or H
+
) will act as an oxidant in the presence of sulphide is accounted for in the lifetime predic-
tions. In fact, corrosion due to sulphide (accompanied by the evolution of H
2
) is predicted to account
for >95% of the total wall loss within the 100,000-yr design life.
In corrosion science, the thermodynamic stability of a material in a given environment is typically
represented in the form of a potential-pH (E-pH) figure, commonly referred to as a Pourbaix diagram
(Pourbaix 1974). Pourbaix diagrams graphically illustrate the regions of thermodynamic stability of
solid and dissolved species as a function of the pH and redox potential (E
H
). The boundaries between
different solids represent the equilibrium conditions for conversion of one phase to another or, in the
case of boundaries between a solid and a dissolved species, the equilibrium for a given activity of
the dissolved component. Of course, the location of the various zones of stability depend not only
on the values of the thermodynamic parameters, but more importantly on the identity of the species
and the reactions between them. The selection of which species and reactions to include is based on
experience of the corrosion characteristics of the given material or alloy in the given environment.
Figure 1-1 shows the E-pH diagram for the Cu-H
2
O system for cases in which the assumed stable
Cu(II) solid corrosion product is either CuO or Cu(OH)
2
. In both cases, the stability of Cu in pure
water is indicated by the fact that the stability field for Cu
2
O lies above the H
2
O/H
2
equilibrium
(denoted as line (a) in the figures).

8 TR-10-69
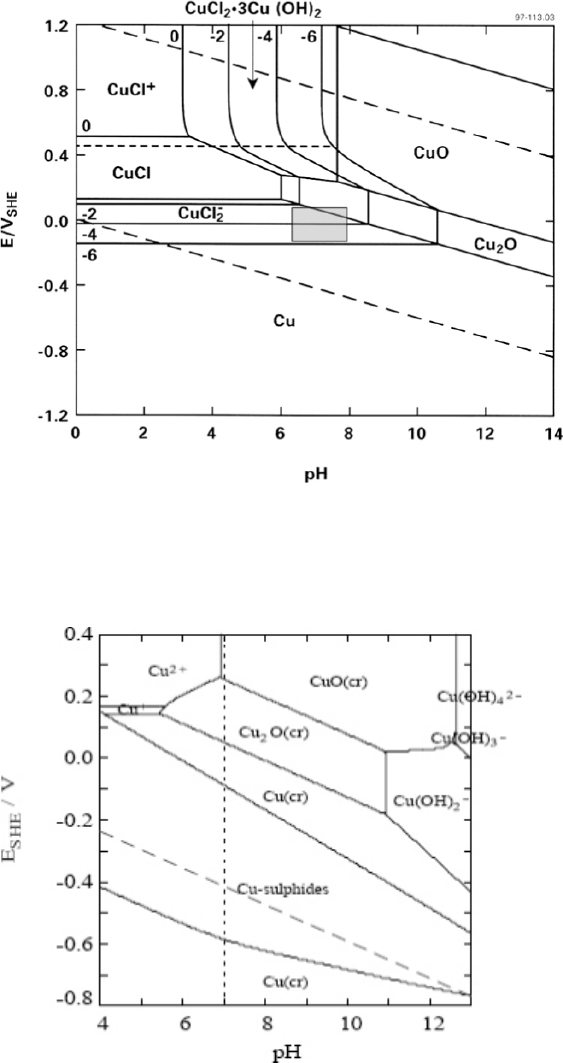
Figure 1-1.
Pourbaix diagrams for the copper-water system at 25°C considering different forms of
copper(II) solid (Pourbaix 1974).
TR-10-69 9
The thermodynamic stability of copper and of the oxides diminishes in the presence of chloride ions
(Figure 1-2). For example, in 1 mol/kg Cl
–
, both CuCl and CuCl
2
·3Cu(OH)
2
are stable at certain pH
and E
H
. Furthermore, Cu becomes thermodynamically unstable in water at low pH, as indicated by
the fact that the Cu/CuCl
2
–
equilibrium line for a dissolved copper activity of 10
–6
mol/kg crosses the
H
2
O/H
2
equilibrium line at pH 2-3 (Figure 1-2).
Copper is also unstable in water in the presence of sulphide ions (Figure 1-3). The stability field
for copper sulphide (primarily Cu
2
S, with possible non-stoichiometric Cu
2–x
S species) straddles the
H
2
O/H
2
equilibrium line at all pH values.
Of course, copper could be thermodynamically unstable in other aqueous systems if there is a stable
corrosion product that suppresses the E-pH range of stability for copper metal. In the case of the
pure water system, however, this would require that this alternative corrosion product would need to
exhibit a stability greater than that of Cu
2
O. Although this is possible it would seem unlikely, since
such a species has not been identified on the basis of the 3,000-plus years of experience of mankind
with copper and copper alloys and the even longer geological timeframes associated with native
copper deposits.
Starting in 1986, Hultquist and co-workers have published a number of articles questioning the
conventional position that copper is thermodynamically stable in O
2
-free pure water (Hultquist 1986,
Hultquist et al. 1989, 2008, 2009, Szakálos et al. 2007, 2008). Other researchers have published
evidence that either contradicts or calls into question the claims of Hultquist et al. (Eriksen et al.
1988, 1989, Johansson 2008, Möller 1995, Simpson and Schenk 1987, Apted et al. 2009). This
report comprises a critical review of the literature on the corrosion of copper by water and a series
of supplemental analyses aimed at providing insight into the issues raised in the published literature.
The critical review in Section 2 includes articles by both Hultquist and co-workers and by those that
dispute their claims. The supplementary analyses in Section 3 include discussion of the formation
and properties of adsorbed Cu-OH species, the evidence for the corrosion of copper in anoxic
Cl
–
solutions, the role of dissolved O
2
in the corrosion of copper, corrosion potential considerations,
and the implications of the proposed instability of copper in water on the long-term performance
of copper canisters in a KBS-3 repository. Finally, in Section 4, a decision-tree approach is used to
assess whether there is evidence for the oxidation of copper by water and, if so, what the impact
would be on copper canisters.
10 TR-10-69
Figure 1-2.
Pourbaix diagram for the copper-water-chloride system at 25°C for a chloride activity of
1 mol/kg (King and Kolar 2000). The shaded box shows the range of corrosion potentials in O
2
-containing
solution.
Figure 1-3.
Pourbaix diagram for the copper-water-sulphur system at 25°C for a total sulphur
concentration of 2·10
–4
mol/kg and a total dissolved copper concentration of 10
–6
mol/kg
(Puigdomenech and Taxén 2000).
TR-10-69 11
2 Critical review of the literature
In this section, the various key studies on both sides of the debate are critically reviewed. Only
documents that are available to the author and in the public domain have been reviewed. The
available information includes not only articles and papers in the peer-reviewed literature, but also
discussion between various parties and information posted to various websites, all of which are
publically accessible.
Figure 2-1 shows a timeline of the articles that have appeared in the scientific literature (with the
exception of the June 1984 article in the Dagens Industri weekly technical journal) and which
represent the key articles reviewed here. Following the publication of some of these articles, there
has been an exchange of comments between the authors of the articles and critics of the work. These
comments and the subsequent rebuttals are also captured here (both those that have been published
in the scientific literature and those that have been posted to various websites), since they represent a
key component of the current debate and of the scientific method of review and critique.
2.1 Critical review
2.1.1 The original article of Hultquist, Corrosion Science 1986
Following an initial newspaper report in 1984, Hultquist published the results of a study of the
corrosion of copper in pure water as a Short Communication in Corrosion Science in 1986. The
paper describes the results of a 1,200-hour experiment in which a number of polished copper strips
(>99.7 wt.%, total surface area 1,460 cm
3
) were exposed to initially aerated deionised, double
distilled water in a conical flask. The flask was apparently sealed using a weight impressing upon an
O-ring gasket. The glass vessel was stored “under dark conditions” in order to exclude photoelectro-
chemical effects. The temperature of the test was 24±2°C.
A solid-electrolyte hydrogen probe was inserted in the headspace above the solution in order to
detect any H
2
evolved from the solution. Prior to the experiment, the probe was calibrated with a
range of H
2
/N
2
gas mixtures and produced the theoretical 60 mV/dec response.
No attempt was made to initially deaerate either the water or the headspace, the volumes of which
were 1.2 dm
3
and 0.02 dm
3
, respectively. The concentration of dissolved O
2
in water in contact with
air at 24°C is 2.72·10
–4
mol·dm
–3
(Battino et al. 1983). The total amount of O
2
in the vessel at the
beginning of the experiment was, therefore, 3.26·10
–4
mol dissolved in the water plus 1.72·10
–4
mol
in the headspace, for a total amount of 5.0·10
–4
mol O
2
. The effectiveness of the weighted O-ring seal
is unknown and it is possible that additional atmospheric O
2
entered the vessel during the course of
the experiment.
At the end of the experiment, a mass gain of 21.2 mg was observed. On the assumption that the mass
change was due to the reaction
2Cu + H
2
O→Cu
2
O + H
2
(2-1)
then the mass change corresponds to the loss of 2.65·10
–3
mol (168 mg) Cu. In addition, it was
estimated that 0.6 mg of dissolved Cu remained in solution, for a total loss of Cu of 169 mg
(2.66·10
–3
mol). Averaged over the entire duration of the experiment, the mean corrosion rate based
on the mass loss is 0.096 µg·cm
–2
·h
–1
(0.94 µm/yr). (Note: there are minor differences between
the masses and rates given here and those reported by Hultquist (1986), presumably as a result of
rounding errors.)

12 TR-10-69
Figure 2-1.
Timeline of key articles and papers on the subject of the corrosion of copper by water.
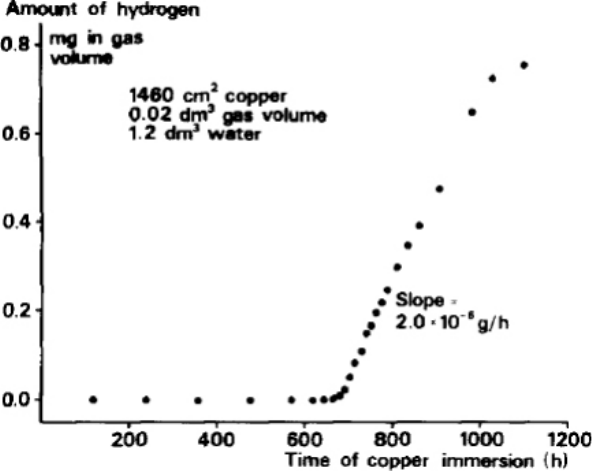
Hultquist (1986) reported the presence of H
2
in the headspace above the water (Figure 2-2).
Accumulation of H
2
was observed after a period of 650 hr. Between that time and the end of the
experiment, approximately 0.8 mg of hydrogen accumulated at a rate (based on the slope of the line
in Figure 2-2) of 2.2·10
–6
g/hr which, based on Reaction (2-1), is equivalent to a corrosion rate of
0.096 µg·cm
–2
·h
–1
(0.94 µm/yr), exactly the same as estimated from the mass loss.
It is interesting to note that the rate of hydrogen production appears to be leveling off towards the
end of the experiment (Figure 2-2), an observation that could have been confirmed if the experiment
had been allowed to continue.
1984
1985
1986
1987
1988
1989
1990
1991
1992
1993
1994
1995
1996
1997
1998
1999
2000
2001
2002
2003
2004
2005
2006
2007
2008
2009
2010
Hultquist
Corros. Sci. 26
, 1986,
173-177
Hultquist et al.
Corros. Sci. 29
, 1989,
1371-1377
Hultquist et al.
Catal. Lett., published
online 28 July 2009
Hultquist et al.
Proc. 17
th
ICC,
2008, paper no. 3884
Szakálos et al.
ESL 11
, 2008, S2-S2
Szakálos et al.
ESL 10
, 2007, C63-
C67
BRITE group
SSM Report 2009:30
Simpson and Schenk
Corros. Sci. 27
, 1987,
1365-1370
Eriksen et al.
Corros. Sci. 29
, 1989,
1241-1250
Johansson
ESL 11, 2008, S1-S1
Möller
SKI Report 95:72
Eriksen et al.
SKB Technical Report
88-17
Hultquist
Dagens Industri,
June 1984
Gråsjö et al.
Mats Sci Forum 185-
188, 1995, 703-712
Seo et al.
Proc. 10
th
ICC 1987
Paper 2.31
Bojinov et al.
Corros. Sci. 52
, 2010,
2917-2927
Korzhavyi and
Johansson
SKB TR-10-30
TR-10-69 13
Based on the similarity between the corrosion rates estimated based on the observed rate of H
2
accumulation and the time-averaged mass loss, Hultquist (1986) proposed that the H
2
observed
was a result of the cathodic reduction of H
+
(or H
3
O
+
) in support of the oxidation of Cu to Cu(I).
Furthermore, it was stated that H
+
(or H
3
O
+
) was the only oxidant and that the O
2
initially present in
the flask was consumed by an interfacial process involving hydrogen atoms produced by the reduc-
tion of H
+
(or H
3
O
+
), in a reaction of the type
4H + O
2
→2H
2
O (2-2)
This is a very important point in Hultquist’s claims which has gone overlooked. Not only does
Hultquist claim that H
2
O is an oxidant for Cu, but he also proposes that H
2
O is reduced prefer-
entially and to the exclusion of O
2
as an oxidant. Notwithstanding the kinetic limitations of both
the reduction of O
2
and of H
2
O on Cu, the thermodynamic driving force for the reduction of O
2
is
1.23 V more positive than that for H
2
O reduction. Not only is the reduction of O
2
thermodynamically
favoured over the reduction of H
+
(or H
3
O
+
), but the initial concentration of dissolved O
2
is over
three orders of magnitude greater than that of H
+
(or H
3
O
+
).
Apart from the observation that H
2
was reported in the flask, it is not clear why Hultquist (1986)
would exclude O
2
as the most likely oxidant for the mass gain observed. It is interesting to note that
the estimated amount (by the current author) of O
2
in the flask initially (5.0·10
–4
mol O
2
) would
result in the corrosion of 127 mg Cu via the reaction:
4Cu + O
2
→2Cu
2
O (2-3)
This amount is less than the 169 mg mass loss reported by Hultquist (1986), but the difference could
be due to air leaking into the flask through the O-ring gasket during the test.
Finally, in recognition of the fact that the amount of H
2
reportedly observed is contrary to thermo-
dynamic prediction, Hultquist suggested that the cathodic reaction is irreversible, implying that
conventional thermodynamics cannot then be used to predict an upper H
2
pressure. The logic of the
argument in this final paragraph of the paper is somewhat unclear.
Figure 2-2.
Time-dependent amount of hydrogen in the headspace above the water as reported by
Hultquist (1986).
14 TR-10-69
The conclusions from the paper were that (Hultqist 1986):
“(1) Hydrogen is evolved during corrosion of copper , i.e. copper is oxidised by water.
(2) The escape rate of hydrogen from the corroding system is of decisive importance for the
corrosion kinetics of copper in water.”
This second conclusion is interesting for a number of reasons. First, there was no discussion of this
issue at all in the body of the text and there was no suggestion that H
2
could escape from the flask.
Therefore, the basis for the conclusion is obscure. Second, regardless of the basis for the conclusion,
it is inconsistent with the suggestion that the H
2
evolution reaction is irreversible. If this reaction is
irreversible, then the partial pressure of H
2
is irrelevant. Finally, if we assume that the conclusion is
correct, then it has implications for the relevance of this proposed mechanism for the corrosion of
copper canisters in the repository (see Section 3.6).
2.1.2 Simpson and Schenk, Corrosion Science 1987
In response to the article by Hultquist (1986), Simpson and Schenk (1987) published a Short
Communication in Corrosion Science describing their own attempts at measuring H
2
due to corro-
sion of copper in aqueous solution. This work was funded by Nagra and was an extension of their
work using the measurement of the rate of H
2
evolution to determine the anaerobic corrosion rate of
C-steel for the Swiss programme. There were a number of differences, and a number of similarities,
between the experimental methods used in both studies. The differences, and their significance, were
discussed by Hultquist et al. (1989) and are summarised in Section 2.1.5. The major experimental
differences between the two studies were that Simpson and Schenk (1987) used a simulated Swiss
ground water and a NaCl solution instead of pure water and the solutions were deaerated prior to the
tests. The significance of the Cl
–
ion is discussed in more detail in Section 3.2.
Simpson and Schenk (1987) used gas chromatography to measure the concentration of H
2
in a N
2
stream passed over the surface of the solutions containing the copper strips. The sensitivity of the
technique to detect H
2
evolved by corrosion is dependent on the N
2
flow rate. With the normal flow
rate of 0.1 dm
3
·hr
–1
, the detection limit was equivalent to a Cu corrosion rate of 3 nm/yr (assuming
corrosion occurs via Reaction (2-1)). In some tests, the gas flow rate was stopped at the end of the
experiment and any H
2
allowed to accumulate under non-flowing conditions for a period of 50 hrs.
The headspace gas was later collected in a known volume of N
2
and again analysed for H
2
by gas
chromatography. The corresponding “detection limit” of this batch technique was one order of
magnitude lower, i.e. a corrosion rate of 0.3 nm/yr.
No H
2
was detected from either the ground water or NaCl solutions at temperatures of 50°C or
80°C using either continuously flowing N
2
or the batch technique.
Small mass gains (0.56–1.0 mg) or mass losses (0.4 mg) were observed in the simulated ground
water and NaCl solution, respectively. (The surface area of Cu was 2,400 cm
2
in tests at 50°C
and 1,200 cm
2
at 80°C.) These mass changes were attributed to corrosion caused by trace levels
of residual O
2
left in the apparatus after inserting the specimens into the pre-deaerated solutions.
The mass gain in the simulated ground water solution could be the result of Cu
2
O formation or the
precipitation of a mineralised film. In the NaCl solution, Cu would dissolve anodically as CuCl
2
–
(King et al. 2001) with minimal precipitation of a solid corrosion product. The gas chromatography
technique was sufficiently sensitive that, had these small mass gains or losses been supported by the
reduction of H
+
(or H
3
O
+
), the resultant H
2
would have been detected.
Simpson and Schenk (1987) were unable to explain the reason for the differences between their
observations and those of Hultquist (1986).
2.1.3 Seo et al. Proc. 10
th
ICC, 1987
Seo et al. (1987) published the results of various experiments in which Cu, Zn, and Fe samples
were exposed to initially aerated water. The apparent production of H
2
was monitored based on
(i) the increase in total pressure (Zn only), (ii) the response of a solid state probe (Zn and Cu), or
(iii) the potential of a Pd electrode immersed in the water (Cu or Fe). Additional experiments were
performed in which a Pt wire immersed in the water was used to monitor the rate of O
2
consumption.

TR-10-69 15
The experiment in which the solid-state electrode was used to monitor the rate of H
2
generation from
copper is the same as that described by Hultquist (1986). Eriksen et al. (1988, 1989) have criticised
the use of this electrode and suggested that the measured voltages were in error. Szakálos et al.
(2007) subsequently recognised the possible error in these measurements, and no further discussion
of these results is given here.
In a second type of experiment, a copper wire was sealed in a glass tube along with water and a thin
Pd sheet. A saturated calomel reference electrode (SCE) was also inserted into the cell in order to
make potential measurements of the Cu wire and Pd sheet, the latter being used as a measure of the
amount of H
2
generated. A similar test was also performed using an Fe wire.
Figure 2-3 shows the time dependence of the potentials of the Pd and Cu electrodes (and the
corresponding data for the experiment with an Fe wire) (Seo et al. 1987). The decrease in potential
for the Cu and Fe wires was taken as a measure of the consumption of O
2
, whilst the potential of the
Pd was taken as a measure of the amount of H
2
absorbed by the sheet. These latter measurements
are questionable, however, because, using the calibration table provided by the authors, there would
appear to be an order of magnitude more H absorbed in the Pd sheet in the Cu experiment than in the
equivalent test with Fe wire, despite a more vigorous evolution of H
2
in the latter.
Figure 2-3.
Time dependence of the potentials of a Pd sheet (cross-in-circle) and Cu wire(open circle)
exposed to initially aerated water sealed in a glass tube (see illustration in the figure) (Figure 4 of Seo
et al. 1987). Corresponding data for an experiment with an Fe wire are shown in the square symbols.
The crosses are the potential of the Pd sheet in a blank experiment with neither Cu nor Fe.
16 TR-10-69
Of more interest is the potential of the Cu wire. The pH of the water measured at the end of the test was
pH 7, whereas in a separate test with Fe an increase in pH to pH 9.3 was measured. The corresponding
equilibrium potentials for the H
2
/H
2
O couple at 1 bar pressure are –0.655 V
SCE
at pH 7 and –0.791 V
SCE
at pH 9.3. Whilst the steady-state potential of the Fe wire (approximately –0.84 V
SCE
) is significantly
below the equilibrium potential, that for the Cu (approximately –0.50 V
SCE
) is ~160 mV more positive.
Since, if H
2
O is the oxidant in this system, the potential of the Cu must be more negative than the
equilibrium potential for the H
2
/H
2
O reaction, the partial pressure of H
2
would have to be <10
–5
bar.
Such a potential value is inconsistent with oxidation of Cu supported by the reduction of H
2
O.
Seo et al. (1987) then proposed a mechanism in which the O
2
in the system, instead of being cathodi-
cally reduced on the copper surface, is consumed by reaction with H atoms produced by the reduc-
tion of water. The reduction of H
2
O is supposed to occur in both aerobic and anoxic conditions. The
onset of H
2
evolution corresponds to the point at which all of the initial O
2
has been consumed by H.
The only evidence provided for this proposed mechanism were results from gas-phase studies with
sub-monolayer coverage by H
2
O and/or O
2
. Hultquist and co-workers also propose this mechanism
in subsequent papers and it is discussed in more detail below.
2.1.4 Eriksen et al. SKB TR-88-17 and Corrosion Science 1989
Eriksen and co-workers also studied the production of H
2
from the corrosion of copper using gas
chromatography in an SKB-funded study that was published both as an SKB report (Eriksen et al.
1988) and as a Short Communication in Corrosion Science (Eriksen et al. 1989). The experimental
arrangement shared similarities with both the original work of Hultquist (1986) and the subsequent
work of Simpson and Schenk (1987). In common with Hultquist’s experiment, Eriksen et al. (1988,
1989) used deionised, double-distilled water with approximately the same volume of water and
surface area of copper of similar quality. (Eriksen et al. also used a higher purity 99.9995 wt.% Cu
in a second test.) In common with Simpson and Schenk’s experimental technique, Eriksen et al.
pre-deaerated both the water and vessels (although incompletely) and used gas chromatography to
detect any H
2
evolved (although with a lower sensitivity to that of Simpson and Schenk). In order
to identify the corrosion products formed on the copper, Eriksen et al. used X-ray photoelectron
spectroscopy (XPS, also known as ESCA) and cathodic stripping voltammetry, the latter technique
also providing a measure of the quantity of reducible corrosion product on the surface.
Eriksen et al. (1988, 1989) did not observe any H
2
produced from the exposure of copper to
deionised, doubly distilled water during the exposure period of 61 days.
Both XPS and cathodic stripping voltammetry identified the corrosion product, which was present
as visible patches on the Cu strip, as Cu
2
O. The time-averaged corrosion rate determined from the
mass loss varied from 0.0082–0.014 µg·cm
–2
·h
–1
(0.08–0.14 µm/yr) for the 99.7 wt.% purity Cu to
0.021 µg·cm
–2
·h
–1
(0.21 µm/yr) for the 99.9995 wt.% Cu. This corrosion was attributed to residual
O
2
in the test vessel following incomplete initial deaeration, a claim which is consistent with the fact
that the highest corrosion rate was observed for the higher-purity Cu, the surface area of which was
only 40% of that of the 99.7 wt.%-purity Cu sample.
Eriksen et al. (1988, 1989) suggested that the reported observation of H
2
by Hultquist (1986) was an
error caused by long-term drift in the output of the solid electrolyte H
2
probe. Szakálos et al. (2007)
have subsequently acknowledged that the probe measurements could be in error.
2.1.5 Hultquist et al. Corrosion Science 1989
The Hultquist et al. (1989) publication in Corrosion Science addressed three areas, namely:
1. a critique of the paper by Simpson and Schenk (1987) based on a discussion of the supposed
significance of some of the differences in experimental technique,
2. a presentation of a simple qualitative experimental illustration of the corrosion of copper by
water, and
3. a description of possible corrosion products of the corrosion of copper in water and the associated
thermodynamic properties.
Hultquist et al. (1989) sought to explain the contradiction in the results from the Hultquist (1986)
and Simpson and Schenk (1987) studies by focusing on the differences in four aspects of the respec-
tive experimental methodologies. These differences and their suggested significance are:
TR-10-69 17
Surface finish of copper. Simpson and Schenk (1987) did not refer to any mechanical polishing of the
copper sheet used in their tests, whereas Hultquist (1986) polished the surfaces to an 800-grit finish.
Hultquist et al. (1989) speculated that the copper used by Simpson and Schenk may have been bright
annealed in a H
2
atmosphere, resulting in absorption of hydrogen into a near-surface layer which,
it was then claimed, would suppress corrosion by the proposed mechanism. There is no evidence
to support the claim by Hultquist et al. (1989) that the copper used by Simpson and Schenk did
indeed contain such an altered surface layer. Furthermore, Hultquist et al. (1989) do not provide any
explanation for how such a surface layer, if it indeed existed, would suppress the corrosion process.
Hultquist et al. (1989) also claimed that by mechanically polishing the copper, Hultquist (1986)
would have produced an oxide-free surface. Mechanical polishing with wet or dry SiC paper does
not produce an oxide-free surface, which is the reason it is necessary to cathodically clean metal
electrode surfaces in situ following polishing and prior to making electrochemical measurements.
Composition of solution. Hultquist et al. (1989) suggest that the increased concentration of dissolved
Cu that would have been present in the Cl
–
solutions used by Simpson and Schenk could have lead
to an increase in pH and the formation of a more-protective surface film, thus explaining the lower
observed corrosion rate. Whilst it is true that the solubility of Cu(I) is increased by the presence of
Cl
–
(due to the formation of Cu(I)-chloro complexes such as CuCl
2
–
), hydrolysis of these species
would, if anything, have lead to a decrease in pH due to reactions such as
2CuCl
2
–
+ H
2
O→Cu
2
O + 4Cl
–
+ 2H
+
(2-4)
Technique for hydrogen detection and presence of oxygen. Hultquist et al. (1989) defended the use of
the solid electrolyte H probe by claiming that, in a study of H
2
evolution from the corrosion of Zn,
the response of the same probe had been validated against manometric measurements of the rate of
pressure increase. Simpson and Schenk (1987) used gas chromatography to detect any evolved H
2
and had not criticised the use of the solid electrolyte H probe used by Hultquist (1986). However,
at the end of a detailed and rather unclear discussion of the functioning of this probe, Hultquist
et al. (1989) questioned Simpson and Schenk’s (1987) conclusion that the presence of 2 ppm O
2
in the flowing N
2
stream in the latter’s experiment would not have suppressed copper corrosion by
water if that reaction were possible. Hultquist et al. (1986) stated that “We do not claim that this [the
suppression of H
+
/H
3
O
+
reduction by 2 ppm O
2
] is the case in Ref. 1 [(Simpson and Schenk 1987)]
but it cannot be ruled out completely.” This seems to be a contradictory argument for Hultquist et al.
(1989) to make because Hultquist (1986) contended that the reduction of H
+
/H
3
O
+
proceeded even
in aerated solution and that, indeed, it was the reaction between H atoms produced by this reduction
reaction and O
2
that was the cause of the consumption of O
2
.
The copper content in solution at the end of the experiment. Hultquist et al. (1989) seem to suggest
that Simpson and Schenk erred in not measuring the dissolved Cu concentration at the end of their
tests. It is not clear how this would have changed the conclusions of Simpson and Schenk (1987).
The simplified experimental technique discussed by Hultquist et al. (1989) comprised polished
copper strips placed in glass tubes initially filled with aerated distilled water and sealed with either
a Pd or a Pt membrane. The concept was that Pd would allow any H
2
produced by corrosion of the
copper to escape and for corrosion to therefore continue. In contrast, H
2
would not permeate through
the Pt membrane and, once an equilibrium pressure of H
2
had built up within the glass tube, corro-
sion would stop. Differences in the extent of corrosion in the two vessels were to be determined by
visual inspection of the degree of corrosion product formation.
Hultquist et al. (1989) carried out tests for a period of 3,000 hr at a temperature of 50°C. Up to
200-hr exposure the copper strips in both tubes changed appearance to a similar degree. During
this period, the authors suggested that the initially trapped O
2
was consumed, although they did not
state via which process (i.e. cathodic reduction or heterogeneous reaction with H atoms produced
by the reduction of H
+
/H
3
O
+
). For the period 200–3,000 hrs, only the copper in the Pd-sealed glass
tube continued to change colour. At the end of the test XPS (ESCA) was used to identify the nature
(mainly Cu
2
O) and, user ion sputtering, the thickness of corrosion product, which was reported to
be 5–6 times greater on the samples from the Pd-sealed tube. The authors claimed that this was
evidence for the corrosion of copper in anoxic water and that the “kinetics of this corrosion can be
ruled by diffusion of hydrogen through the enclosure of the system.”
SIMS was used to characterise the corrosion products on the samples, as well as on copper samples
exposed to continuously aerated water. The higher relative abundances of ions containing H from the
samples in the sealed glass tubes was taken as evidence that H
+
/H
3
O
+
had been reduced during the tests.
18 TR-10-69
It is difficult to comment on the significance of these results without having been present during the
experiments themselves. Although the explanation inferred by Hultquist et al. (1989) is plausible,
more corrosion could have occurred in the Pd-sealed glass tube simply because this seal failed
during the test, whereas the Pt seal did not (or did not to the same degree). Neither is the SIMS
evidence convincing. Many different types of secondary ions are produced during the Ar
+
-ion
bombardment of the surface. The differing amounts of H in the products could simply represent
different degrees of hydration of the corrosion products formed. We note that the sealed glass tube
experiments and the experiment in continuously aerated water were performed at different tempera-
tures; at temperatures of 50°C and room temperature, respectively. Figueroa et al. (1986) studied
the temperature dependence of the film breakdown (pitting) potential (E
B
) of copper in Cl
–
solutions
at pH 9 and observed a minimum value at a temperature of 15°C, with the value of E
B
shifting to
more-positive values with increasing temperature up to the maximum temperature of 55°C used in
their studies. Figueroa et al. (1986) attributed this increasing protectiveness of the film to differing
degrees of hydration over the temperature range studied.
Hultquist et al. (1989) briefly describe a second series of experiments in which the upper surface of
the Pd seal was contacted by a column of water into which a saturated calomel reference electrode
was inserted. Electrical contact with the upper surface of the Pd membrane was made with a Pt wire.
This arrangement was designed to act as a crude hydrogen electrode, with the measured potential
serving as a qualitative measure of the amount of hydrogen permeating through the Pd membrane.
Experiments were set up with Zn, Fe, and Cu samples and a blank test without any metal. The most-
negative potential was observed in the test with the Zn samples, followed by the Fe samples, with
the least-negative potential recorded with Cu.
As with the glass tube experiments described above, it is difficult to criticise the reported observa-
tions without a more-detailed knowledge of exactly how the experiments were conducted.
Hultquist et al. (1989) conclude the article with a discussion of possible alternative corrosion
products. The underlying thesis of this discussion was that the corrosion products of the corrosion of
copper in pure water are more complex than those shown in Reaction (2-1) and that thermodynamic
predictions based on that stoichiometry are not valid. The authors present the results of SIMS analy-
sis of Zr, Zn, Fe, and Cu surfaces exposed to H
2
, O
2
, or H
2
O at a partial pressure of ~10
–5
Pa, report-
ing species such as CuH
+
, CuO
+
, and CuOH
+
. Based on the relative amounts of these species for the
different exposure conditions (and of the equivalent species for Zn and Fe), it was concluded that
CuH
+
is formed to a greater degree in the presence of H
2
O than when O
2
is present. Presumably the
inference was meant to be that H
2
O is reduced on copper, especially in the absence of O
2
. However,
the exposure of Cu to these conditions has no relevance for the behaviour of copper in anoxic H
2
O.
Aqueous corrosion is an electrochemical process that requires a minimum of several monolayers of
water to occur. The formation of a sufficiently thick water layer is associated with the well-known
critical relative humidity for atmospheric corrosion of 60–80% relative humidity. At a temperature of
25°C, this critical %RH corresponds to a water vapour pressure of ~2 kPa, approximately 8 orders of
magnitude greater than the pressure in the tests from which these corrosion products were identified.
Based on this and additional evidence from UHV studies (Spitzer and Lüth 1985) and from studies in
solution (Seo et al. 1987), Hultquist et al. (1989) list a series of mass- and charge-balanced reactions that
they propose account for the corrosion of copper in H
2
O (with or without O
2
). These reactions involve the
formation of a CuOH or Cu
2
OH intermediate species and the cathodic reduction of H
+
to form atomic H:
Cu + H
2
O→Cu(H
2
O)
ads
→CuOH+H (2-5a)
followed by
2CuOH→Cu
2
O + H
2
O (2-5b)
or
CuOH→CuO+H (2-5c)
in the case of the CuOH intermediate, and
2Cu + H
2
O→2Cu(H
2
O)
ads
→Cu
2
OH + H (2-6a)
followed by
Cu
2
OH→Cu
2
O + H (2-6b)
in the case of the Cu
2
OH intermediate.
TR-10-69 19
It should be emphasised here that much of the evidence for these intermediate species comes from
studies in UHV conditions with very low H
2
O vapour pressure.
There is no role in these reactions for the dissolved O
2
present in solution initially in the glass tubes.
Hultquist et al. (1989) claim that, instead of being cathodically reduced on the Cu surface, the O
2
is
consumed by reaction with the H formed in Reactions (2-5a), (2-5c), (2-6a), or (2-6b), via
H + ½O
2
→OH (2-7a)
or
2H + ½O
2
→H
2
O (2-7b)
In addition to the consumption of O
2
, atomic H can also (i) combine and evolve as H
2
, (ii) enter the
copper, or (iii) escape from the system via some ill-defined pathway involving “surface diffusion
and/or desorption.”
This suggestion that O
2
does not directly participate in the corrosion of Cu in aerated solutions is
unconventional but is consistently stated in Hultquist’s publications (Hultquist 1986, Gråsjö et al.
1995, Seo et al. 1987, Szakálos et al. 2007). This issue is analysed in more detail in Section 3.4.
2.1.6 Möller, SKI Report 95:72
Möller (1995) attempted to replicate the “glass tube” experiments reported by Hultquist et al. (1989).
Tests were performed in both quartz and soda glass test tubes sealed with either Pd or Pt membranes
using initially aerated pure water and heated to a temperature of 50°C, as in the original study, for a
period of 2 years. The tests in the quartz tubes were done in triplicate.
Möller’s (1995) observations were quite different from those of Hultquist et al. (1989). In particular,
Möller (1995):
• didnotobserveanyvisualdifferenceinthecolourofthecopperinthePd-andPt-sealedtest
tubes,
• didnotobserveanysignificantdifferenceintheamountofoxideformedinthePd-andPt-sealed
test tubes, and
• didnotobserveanyoxidegrowthafterthefirstyear,whichMöllerconcludedwasbecauseallof
the oxidant (O
2
) had been consumed.
Möller (1995) also reported that the amount of oxide formed was not exactly equivalent to the
estimated initial amount of O
2
present. He also reported that a significant amount of water had
“disappeared” from the quartz test tubes during the test.
The significance of this latter observation is unclear. The water could have been lost because of
improper seals or by incorporation into a hydrated corrosion product. Alternatively, H
2
O could have
been reduced in the corrosion of copper as proposed by Hultquist and co-workers, although this
suggestion is inconsistent with Möller’s other observations.
20 TR-10-69
2.1.7 Gråsjö et al. Mater. Sci. Forum 1995
Gråsjö et al. (1995) studied the consumption of O
2
and the production of H
2
during the corrosion
of Al, Zn, Fe, Cu, and Grade 316 stainless steel exposed to pure water. The exact experimental
procedure is not clear from the description provided, but it appears that the water was initially
aerated but that the air above the water was removed in some tests, but not in others. Regardless,
the time dependence of the amount of O
2
and H
2
in the headspace above the water was determined
by bleeding off a small volume and analysing it in a separate chamber using mass spectrometry.
Oxygen was consumed during the tests for all the metals except the stainless steel, for which the
corrosion rate was considered too small to significantly consume O
2
, although the sensitivity of the
technique was reported to be equivalent to a corrosion rate of 0.01 µm/yr. Hydrogen was detected
in the case of Al, Zn, and Fe but, interestingly, not for Cu. In the Cu test, approximately 99% of
the original O
2
had been consumed by the end of the 200-hr test but there appears to have been no
attempt to extend the duration of the test to determine if H
2
would have been produced once the
water had become anoxic (as there was in the case of Fe, the test for which was allowed to continue
for 2,000 hrs). Based on the time-dependence of the O
2
concentration, the rate of O
2
consumption in
the Cu test was found to be first order with respect to [O
2
].
Gråsjö et al. (1995) concluded that the first-order kinetics for O
2
consumption was evidence that O
2
was consumed by reaction with H on the metal surface, presumably formed by the reduction of H
2
O.
The authors appear to have discounted the more-conventional explanation that O
2
was consumed by
cathodic reduction on the copper, a reaction that is known to be first order with respect to [O
2
] (King
et al. 1995a, b, Vazquez et al. 1994a, b). The only evidence that Gråsjö et al. (1995) provide in sup-
port of their proposed mechanism are results from gas phase studies at temperatures of 160–400°C in
which no liquid water was present (RH <0.02%) (Hultquist et al. 1993, 1994).
2.1.8 Szakálos et al. Electrochem. Solid-State Letters 2007
The article by Szakálos et al. (2007) has resulted in much discussion about the claims of Hultquist
and co-workers that H
2
O is an oxidant for copper in anoxic water. The paper is an attempt to produce
a more-rigorous study of the proposed phenomenon to address concerns expressed by Eriksen
et al. (1989), and admitted by Szakálos et al. (2007), regarding the validity of Hultquist’s (1986)
H
2
measurements using the solid electrolyte probe. In this latest work, the flux of H
2
passing from
the corrosion cell through a Pd membrane was measured using an ion pump or, alternatively, the
accumulation of H
2
was measured as a pressure increase in a fixed volume on the outlet side of the
membrane.
Experiments were performed using polished oxygen-free high-conductivity (OFHC) Cu in deionised
water for periods of up to 2,300 hrs and at temperatures that were varied between 8°C and 85°C.
Attempts were made to deaerate the water but inevitably trace amounts of O
2
remained in the
system. Further details of the experimental procedure are given by Szakálos et al. (2007).
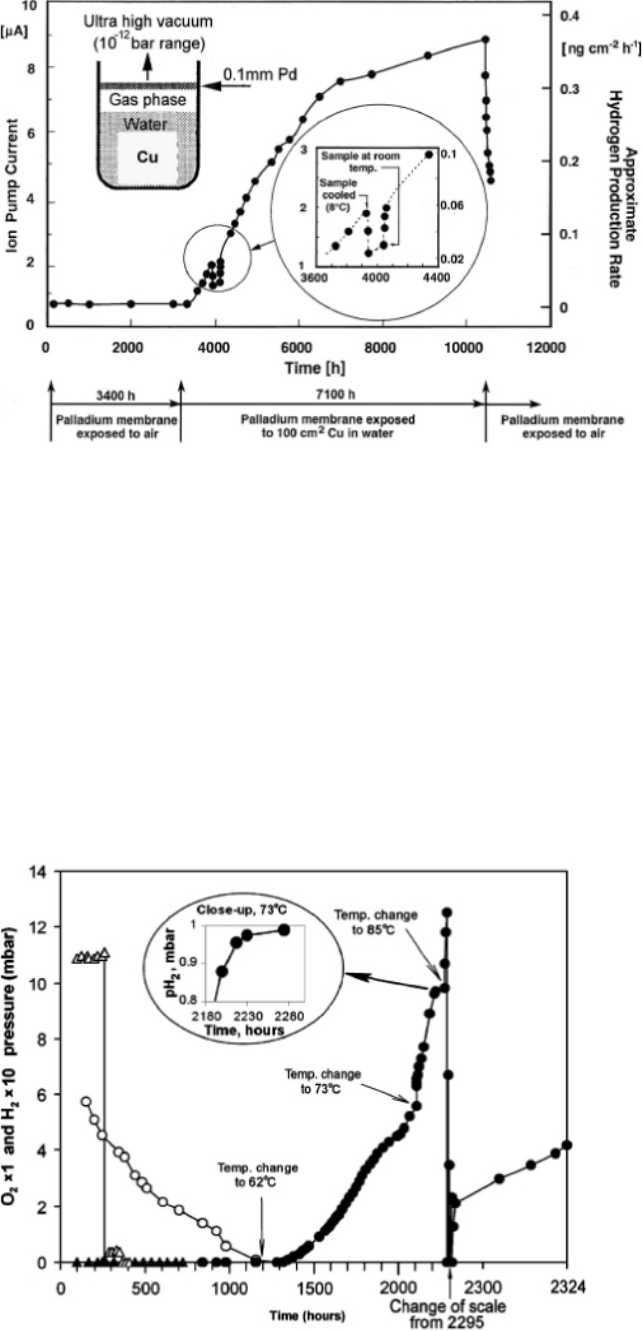
Figure 2-4 shows the results of the copper corrosion experiment in which the ion pump was used to
detect the flux of H
2
exiting the Pd membrane. Szakálos et al. (2007) state that the ion pump current
is proportional to the flux of H
2
, which they also show on the figure. A H
2
flux of 0.4 ng·cm
–2
·hr
–1
is equivalent to a corrosion rate of 0.25 µm·yr
–1
(based on Reaction (2-1)). In some literature, the
ion pump current is said to be a measure of the gas pressure, not the flux of gas. This would make
more sense because, otherwise, the data in Figure 2-4 indicate that the corrosion rate is increasing
continuously with time between 3,400 hr and 10,000 hr. It would also indicate that the H
2
was being
evolved (albeit at a constant rate) whilst the copper was exposed to air during the first 3,400 hr since
a finite ion pump current is measured during this period. The current author knows of no corrosion
system in which the rate of general corrosion increases by a factor of 10 or greater, as is suggested
by the right-hand axis in Figure 2-4.
TR-10-69 21
Figure 2-4.
The reported flux of H
2
produced by the corrosion of copper by water exiting a Pd membrane
as measured using an ion pump (Figure 2 from Szakálos et al. 2007).
Figure 2-5 shows the results of the experiment in which the pressure of H
2
accumulating above the
Pd membrane was measured as a function of time, during which the temperature was increased on
several occasions. Since this is a direct measure of the pressure, the rate of corrosion is given by the
slope of the curve. Hydrogen was not specifically identified in this test and it was simply assumed
that a decreasing pressure represented the consumption of O
2
(open symbols) and an increasing
pressure represented the production of H
2
(full symbols). Following a period of 1,280 hrs, the
pressure was observed to increase. The slope of this curve is relatively constant between changes
in temperature, suggesting a constant corrosion rate at a given temperature. This is in contrast to
the apparent acceleration in the corrosion rate represented by the data in Figure 2-4 and calls into
question the interpretation of the results of these two experiments.
Figure 2-5.
The reported change in pressure due to H
2
produced by the corrosion of copper in water
accumulating above the Pd membrane (Figure 3 from Szakálos et al. 2007).

22 TR-10-69
Another point of interest in Figure 2-5 is the apparent arrest in the increase in pressure at a
temperature of 73°C after ~2,250 hrs (highlighted as an insert in the figure). Szakálos et al. (2007)
interpreted this arrest as indicating that the system was reaching an equilibrium pressure, which they
estimated to be about 1 mbar (101 Pa) at 73°C.
Following the experiment, the corrosion products were examined using powder XRD and SIMS.
The SIMS analysis showed a range of ionic products with varying O and H contents. A similar
trend to that reported by Hultquist et al. (1989) was observed, in which the H content was higher in
tests performed in the absence of O
2
. The powder XRD suggested the presence of CuO and Cu
2
O.
Nevertheless, the authors suggested that the major corrosion product was H
x
CuO
y
, an unspecified
hydrogen- and oxygen-containing Cu species of undefined stoichiometry. Alternatively, the ionic
products observed by SIMS could have resulted from a hydrated Cu
2
O film.
Szakálos et al. (2007) presented a modified Pourbaix (E-pH) diagram for the Cu–H
2
O system that
includes the proposed stability field for the presumed corrosion product H
x
CuO
y
(Figure 2-6).
Comparison with Figure 1-1 shows that the authors have assumed an equilibrium between H
x
CuO
y
and Cu
2
O, with the stability field of the new compound extending to a potential given by the water
reduction line for a H
2
pressure of ~1 mbar (101 Pa) corresponding to the proposed equilibrium H
2
pressure estimated from Figure 2-5.
Based on the proposed mechanism and the estimated field of stability of the proposed new species
H
x
CuO
y
, Szakálos et al. (2007) conclude that copper will corrode in anoxic water due to the reduc-
tion of H
2
O if the H
2
pressure is less than approximately 1 mbar (at 73°C). Hydrogen evolution is
only detectable once O
2
has been removed from the system and, although it is not specifically stated
in this paper, it is implied that any O
2
present is consumed by reaction with atomic H rather than by
direct reduction on the copper surface.
There was some discussion of the results presented by Szakálos et al. (2007) between the authors
and Drs. Hannu Hänninen and Willis Forsling of the Swedish National Council for Nuclear Waste
(Hänninen and Forsling 2007, Szakálos and Hultquist 2008). Among other comments, Hänninen
and Forsling (2007) proposed a number of alternative causes for the H
2
observed by Szakálos et al.
(2007), including: other cell components (such as the stainless steel components, glass, or sealing
materials), desorption of hydrogen absorbed in the material during processing, or the oxidation of a
pre-existing Cu
2
O film or of a Cu
2
O film formed by reaction with residual O
2
to CuO.
The reply by Szakálos and Hultquist (2008) addressed these and other comments that had passed
between the two groups. In response to the latter suggestion regarding the oxidation of a pre-formed
Cu
2
O film, Szakálos and Hultquist (2008) claimed that there was insufficient residual O
2
in the
vessel to produce sufficient Cu
2
O to account for the amount of H
2
observed. However, this response
ignores the air-formed oxide that would have been present on the surface following polishing and
which was not subsequently removed prior to the experiment.
Figure 2-6.
Modified Pourbaix (E-pH) diagram including the proposed stability field for the new species
H
x
CuO
y
(Figure 4 from Szakálos et al. 2007).
TR-10-69 23
2.1.9 Johansson, Electrochem. Solid-State Letters 2008
Johansson (2008) published a comment on the Szakálos et al. (2007) article in which he provided an
alternative explanation for the experimental observations. His major criticisms were that:
1. There is no evidence, in the form of a copper-based mineral for example, for the existence of a
stable H
x
CuO
y
species, unlike the widespread evidence for Cu
2
O and CuO.
2. The H
2
observed is consistent with the corrosion of the stainless steel reaction chamber and the
thickening of the pre-existing passive oxide film.
3. The mass gain exhibited by the Cu can be explained solely in terms of the O
2
initially in the test,
contrary to the claim by Szakálos et al. (2007) that at least 50% of the mass change was the result
of oxygen incorporated into the film due to the reduction of H
2
O.
Szakálos et al. (2008) responded to Johansson’s criticisms and maintained their original position,
stating that:
1. Hydrogen was also observed in the “ion-pump” experiment, in which there was no exposed stain-
less steel surfaces present. Furthermore, even in the “pressure-gauge” experiment where stainless
steel surfaces were exposed to the water, they doubted whether the corrosion rate would have
been sufficient at the temperature of the experiments to account for the amount of H
2
reported.
2. Based on their calculations, there was insufficient initial O
2
to account for the mass gain observed
on the copper.
3. If the conventional E-pH diagram is correct, then Cu
2
O should have been reduced in the presence
of H
2
which did not occur so, by inference, there must be an alternative corrosion product present
that is stable in the presence of H
2
.
The question of the amount of O
2
initially present is a key issue and is, unfortunately, difficult to
resolve. If, indeed, there was more oxygen in the corrosion product than was present initially as O
2
then some or all of the oxygen is most likely to have resulted from the reduction of H
2
O. However,
O
2
is notoriously difficult to remove from aqueous solutions (Butler et al. 1994), as well as being
difficult to prevent from re-entering the experimental apparatus.
In their response, Szakálos et al. (2008) present additional information regarding the hydrogen content
of the copper foil. They suggest that, during the course of the experiment, the H content of the copper
foil increased from 1 wppm to “about one order of magnitude more hydrogen in the copper metal than
in the gas phase”, although Szakálos et al. (2008) do not actually state how much H was in the metal.
The solubility of hydrogen in copper is very small (San Marchi 2006). Measurement of low concen-
trations of H in metals is complicated by artifacts due to hydrated corrosion products and adsorbed
water. Based on extrapolation of the solubility data compiled by San Marchi (2006), the lattice H
concentration in copper at 73°C is of the order of 10
–5
wppm for a H
2
pressure of 1 mbar (101 Pa).
Although some H could also be trapped in the metal due to dislocations and other types of trap,
such a large discrepancy with the reported value of 1 wppm is difficult to explain. Copper hydride is
known, but is unstable at temperatures >0°C (Fitzsimons et al. 1995).
2.1.10 Hultquist et al. 17
th
Int. Corrosion Congress 2008
Further evidence for the corrosion of copper in anoxic water was presented in a wide-ranging paper
by Hultquist et al. (2008). The major focus of the paper was the results from two studies in which,
again, differences were observed in the corrosion behaviour of copper in water in glass vessels
sealed using either Pd or Pt membranes.
In one series of tests, strips of OFHC copper were exposed to water in sealed conical flasks for a
period of 15 years (Figure 2-7). Visually, the copper in the Pd-sealed vessel was covered in a rela-
tively thick black precipitate with distinct areas of green colouration indicative of Cu(II). In contrast,
the copper strips in the Pt-sealed flask, from which H
2
could not escape, appeared to be barely
corroded. This was seen by Hultquist et al. (2008) to be further evidence that H
2
O is an oxidant for
copper and that, because the H
2
evolution reaction is reversible, corrosion will continue if the H
2
is
allowed to escape (as in the Pd-sealed vessel). Hultquist et al. (2008) also noted that the water level
had decreased in the Pd-sealed flask, taking this as further evidence for the reduction of H
2
O.

24 TR-10-69
Hultquist et al. (2008) made a number of other interesting observations on the copper strips from
the Pd-sealed vessel. First, metallographic cross sections through the green-coloured areas showed
evidence for localised attack of the copper (Figure 2-8). The localised attack does not exhibit the
hemi-spherical shaped pits typical of the pitting of copper in fresh water, but appears more like
intergranular attack with selective oxidation of individual grains. A maximum depth of localised
penetration of 70 µm was observed, equivalent to a maximum penetration rate of 5 µm/yr. As
pointed out by Hultquist et al. (2008), this rate is higher than the rate of corrosion in O
2
-containing
environments (King et al. 2001). There are similarities between the observed localised attack with
so-called “ant’s nest corrosion” (Corbett and Elliot 2000, King et al. 2001), but without the tunneling
and inter-linking typical of this form of corrosion found in aerated environments.
The second interesting observation was a quantitative analysis of hydrogen apparently absorbed
by the metal, a phenomenon also referred to in the comment by Szakálos et al. (2008). Hultquist
et al. (2008) show data for the thermal desorption of H from as-received Cu, corroded Cu without
corrosion product, and corroded Cu with corrosion product. Large quantities of H (20 wppm to
>140 wppm) were seemingly released from the corroded copper, with the higher amount from that
with adherent corrosion product.
Figure 2-7.
Appearance of copper strips after exposure to distilled water for 15 years at room temperature in
Pd-sealed (left-hand side) or Pt-sealed (right-hand side) conical flasks (Figure 1 in Hultquist et al. (2008)).
Figure 2-8.
Cross section through a green-coloured region from one of the copper strips exposed to water
in the Pd-sealed flask from Figure 2-7 (Figure 2 of Hultquist et al. 2008).
TR-10-69 25
The third interesting observation, and one which Hultquist et al. (2008) associated with the presence of
absorbed H, was that it was claimed that the mechanical properties of the material had been degraded
by exposure to water. This claim was based on the observation that a crack initiated when a corroded
strip was bent by 180° and a report in the literature that cathodic charging of copper reduces the tensile
strength and strain-to-failure (Panagopoulos and Zacharopoulos 1994). However these latter tests were
conducted under such severe charging conditions (current densities of 10–150 mA·cm
–2
in 2.6 wt.%
H
2
SO
4
containing 10 mg/L As
2
O
3
) that the relevance to the conditions here is questionable (even if
we assume that H
2
O is reduced on the copper surface). For OFHC Cu in contact with 69 MPa gaseous
H
2
, there are conflicting reports of the effect of hydrogen on the mechanical properties of copper, with
some studies showing no effect and others indicating a loss in tensile strength (San Marchi 2006).
Finally, Hultquist et al. (2008) reported the results of SIMS analyses of corrosion products on copper
rods that had also been exposed to water in Pd- or Pt-sealed glass vessels. As previously reported
(Hultquist et al. 1989, Szakálos et al. 2007), higher proportions of H-containing secondary ions were
found for corroded Cu exposed to water in the Pd-sealed vessel. Hultquist and co-workers use these
observations of H-containing corrosion products as evidence that H
2
O is acting as an oxidant, partly
because Pourbaix diagrams conventionally show H-free Cu
2
O and CuO as the stable corrosion prod-
ucts of Cu in H
2
O (even though Pourbaix (1974) also shows an E-pH diagram with Cu(OH)
2
as the
stable Cu(II) corrosion product). However, it has been known for some time that an adsorbed Cu(I)
hydroxide species (CuOH
ADS
) is a precursor of Cu
2
O formed during electrochemical experiments
(King 2002, King et al. 2001). The formation and properties of these species are reviewed in more
detail in Section 3.1. Furthermore, all passive films are hydrated to some degree and those formed
on noble metals, including Cu, are often described as being hydrous in nature (Burke and Murphy
2001, Burke et al. 1990). Therefore, the presence of H in corrosion product films on copper is not
necessarily an indication of the reduction of H
2
O.
2.1.11 Report from the SSM BRITE group 2009
The Barrier Review, Integration, Tracking, and Evaluation (BRITE) group of the Swedish Radiation
Safety Authority (SSM) published a critical review of the Szakálos et al. (2007) article in 2009
(Apted et al. 2009). The review was critical both in the sense that it presented an in-depth analysis of
the original work, supplemented by additional analyses of their own and in the sense that the BRITE
group were skeptical of the relevance to the KBS-3 concept of the reported observations, even if
these findings could be shown to be valid. Although the review was officially published in 2009, a
draft version had been available for review and comment, including by Hultquist and co-workers,
prior to this date.
As noted above, the BRITE group cast doubt on the relevance of the findings of Hultquist and
co-workers, concluding that:
“Based solely on our analyses of the information published up to and including Szakálos et al.
(2007), and from the perspective of the performance of the KBS-3 concept, it could be reasonably
argued that no further work was warranted on the postulated copper corrosion process. …”
The BRITE group performed supplemental analyses of the data presented by Szakálos et al. (2007),
focusing on four possible alternative hypotheses for the generation of H
2
in both the “ion-pump” and
“pressure-gauge experiments” (Apted et al. 2009). It is important to note that the BRITE group did
not dispute the fact that H
2
was generated in these tests. The four alternative hypotheses were:
1. The mechanism proposed by Szakálos et al. (2007) in which H
2
O acts as an oxidant for Cu, with
some of the resultant hydrogen evolving as H
2
gas.
2. Oxidation of a Cu
2
O film (either pre-formed in air or formed due to corrosion by residual O
2
)
to either CuO or Cu(OH)
2
coupled to the reduction of H
2
O, with some of the resultant hydrogen
evolving as H
2
gas.
3. A surface catalysed process resulting in the reduction of H
+
(or H
2
O) to H
2
and the oxidation
of dissolved Cu(I) (formed by the oxidation of Cu by residual O
2
) to dissolved Cu(II).
4. The H
2
gas could have formed from H absorbed into the copper during the original
manufacturing process.
26 TR-10-69
A fifth alternative hypothesis, that the H
2
resulted from corrosion of the stainless steel surfaces
exposed in the experiments (as proposed by Johansson (2008)), was considered to have been
adequately addressed by Szakálos et al. (2008) and was not considered further by the BRITE group.
Supplementary analyses were performed in an attempt to identify which one of these alternative
hypotheses, if any, could best account for the experimental observations. First, an analysis based on a
conventional thermodynamic approach was carried out, which reaffirmed that H
2
O could not oxidise
copper with the formation of Cu(OH)
2
(the formation of Cu
2
O was not considered because of the
assertion by Szakálos et al. (2007) that the corrosion product contains H and is of the form H
x
CuO
y
).
Cupric hydroxide corresponds to the solid species proposed by Szakálos et al. (2007) for the case
x = y = 2. A similar analysis was also performed for the reaction
2Cu + 2H
2
O→2CuOH+H
2
(2-8)
with CuOH corresponding to the case of x = y = 1. This analysis also indicated that CuOH was
thermodynamically unstable with respect to Cu metal for the conditions of the tests. Finally, the
direct oxidation of Cu to Cu
2+
by H
+
Cu + 2H
+
→Cu
2+
+ H
2
(2-9)
is only thermodynamically favourable (at pH 6.5 and 73°C) for [Cu
2+
] < 10
–19
mol·dm
–3
, a condition
that was deemed to be highly unlikely in water in contact with copper. It was concluded, therefore,
that the available thermodynamic evidence did not support the contention of Szakálos et al. (2007)
(Hypothesis 1), but the BRITE group did not entirely rule out the possibility that the proposed
H
x
CuO
y
might exist.
The BRITE group also analysed the apparent time dependence of the corrosion rate from the
ion-pump and pressure gauge experiments. As noted above, the H
2
production (corrosion) rate in the
ion-pump experiment increased with time. Acceleration of the rate of reaction is characteristic of
an autocatalytic process (but is not characteristic of a catalytic process as suggested by the BRITE
group) in which one of the products is the catalytic species. None of the proposed hypotheses
above involve an autocatalytic process. Apparently Szakálos and Hultquist have suggested that the
increasing reaction rate is a consequence of a deteriorating surface film, but the cited Szakálos and
Hultquist (2008) is not listed in the references in the BRITE report.
Next, the BRITE group compared the H
2
generation rates from the two experiments (the ion-pump
and pressure-gauge tests) (Apted et al. 2009). For the ion-pump experiment, a mean corrosion rate of
0.08 µm/yr was derived, which is a factor of three less than the peak rate of 0.23 µm/yr based on the
maximum reported H
2
generation rate of 0.37 ng·cm
–2
·h
–1
(Figure 2-4). Based on the integrated cor-
rosion rate, a pre-existing Cu
2
O-layer thickness of 0.21 µm would be required if H
2
was generated
via (Hypothesis 2)
Cu
2
O + 3H
2
O→2Cu(OH)
2
+ H
2
(2-10)
This oxide film thickness was deemed to be possible since O
2
was initially present in the test vessel.
Interestingly, the corrosion rate estimated by the BRITE group for the pressure-gauge experiment
was approximately 40 times lower than that in the ion-pump experiment, even though the test was
done at a higher temperature. This difference cannot be explained by the attainment of equilibrium in
the pressure-gauge experiment since this condition was only established towards the end of the test.
The BRITE group also estimated activation energies for the two tests and found that they differed.
However, it is difficult to draw firm conclusions from the limited data available.
It was not possible to determine the validity of Hypothesis 3 (the surface-catalysed reduction of H
+
)
based on the available data. Hypothesis 4 (the release of H in the as-received Cu strips) was consid-
ered unlikely because the amounts of H
2
generated exceeded the amount likely present in the metal
by two orders of magnitude (in the case of the ion-pump experiment).
The BRITE group were critical of the solid-phase analyses that had been performed (Apted et al.
2009). They did not consider the SIMS data reliable for a number of reasons, and suggested
that the inclusion of measurements on standard compounds would improve confidence in these
measurements. They were similarly skeptical of the ability of powder X-ray diffraction to adequately
TR-10-69 27
characterise the corrosion products and suggested alternative X-ray techniques that should be used.
Finally, they criticised the “vague and imprecise terms” in which Szakálos et al. (2007) described the
properties of the proposed H
x
CuO
y
solid phase.
Having examined the basis for the claims of Szakálos et al. (2007) based on the scientific evidence
provided, the BRITE group then considered the relevance and implications for the repository system.
Much of this discussion was based on evidence from natural analogues. It was argued that, although
there are a significant number of native (i.e. metallic) copper deposits known, there is no evidence
for Cu ores bodies containing this previously unknown H
x
CuO
y
species as would be expected if this
phase were thermodynamically stable. For example, there is no evidence for alteration rinds on the
surface of copper deposits as would be expected if Cu was slowly altered to H
x
CuO
y
. Neither can it
be argued that H
x
CuO
y
is metastable with respect to Cu (i.e. H
x
CuO
y
was formed initially but it has
since been reduced to Cu metal), since mineralogical and crystallographic evidence indicates that
these deposits were formed as Cu metal. There is no evidence from the geological record, therefore,
for either the formation or the persistence of the proposed H
x
CuO
y
phase.
Finally, the BRITE group considered the implications of Szakálos et al.’s (2007) claims for the
lifetime of the copper canisters and the overall safety of the KBS-3 system. They estimated a canister
lifetime of 625,000 yrs based on their estimated corrosion rate of 0.08 µm/yr, although it could be
argued that the lifetime would only be 217,000 based on the peak H
2
generation rate presented in
Szakálos et al. (2007). The BRITE group argued that even if the canister lifetime was of the order
of 625,000 yrs it would have little impact on safety (as measured by the release of dose-contributing
radionuclides) because the half-lives of the highly soluble radionuclides (e.g. Cl-36, I-129, Cs-135)
are so long that the suggested reduction in the period of containment has little effect on dose.
The overall conclusions from the BRITE group report were that:
• TheinformationpresentedinSzakálosetal.(2007)isincompleteanddoesnotsubstantiatethe
claim that water oxidises copper.
• However,thepossibilitythatthisreactionispossiblecannotbeexcludedbecausenootherviable
mechanism has been demonstrated.
• TheevidenceprovidedbySzakálosetal.(2007)insupportoftheformationofH
x
CuO
y
is
insufficient.
• TherearealternativemechanismstoexplaintheobservationofH
2
formation, but there is
insufficient information to adequately test these hypotheses.
• ThereisnoevidencefromnaturalanaloguesfortheformationorpersistenceofH
x
CuO
y
.
• EveniftheclaimsofSzakálosetal.(2007)arecorrect,thereislittleornoimpactonthesafety
of the KBS-3 disposal system.
• Whilsttheywereinclinedtosuggestthatnofurtherinvestigationofthisissueiswarranted,the
BRITE group recommended than an independent study into the claims be performed under well-
controlled conditions.
2.1.12 Hultquist et al. Catal. Lett. 2009
The latest paper in the series from Hultquist and co-workers (Hultquist et al. 2009) is the first in
which the authors attempt to make predictions of the lifetime of copper canisters in a repository. The
paper is a collection of various topics, including (i) the results of molecular dynamics simulations
of proposed surface species, (ii) further measurements of H
2
evolved from copper corrosion experi-
ments in water, and (iii) predictions of the extent of copper corrosion on copper canisters in the
repository.
A theme that carries through many of the publications reviewed here, and which continues in
Hultquist et al. (2009), is the reliance by Hultquist and co-workers on information derived from
studies of copper surfaces exposed to gaseous water vapour and/or O
2
at low pressures. These gas-
phase studies have been used to justify the proposed consumption of O
2
by H atoms formed from
the reduction of H
2
O in aqueous solution and the formation and existence of H-containing corrosion
products. The fundamental conditions and the processes that operate in the gas phase are totally
28 TR-10-69
different from those that occur in solution. In solution, ionic species are hydrated and the interface
comprises a double-layer structure that affects, among other processes, the adsorption of species on
the surface and the nature of charge transfer reactions. Electrochemical charge-transfer processes are
responsible for oxidation and reduction processes at the surface. The use of information gathered from
gas phase studies, in which neither a double layer exists nor electrochemical reactions occur, must be
considered tenuous at best.
The molecular dynamics simulations were an attempt to obtain a value for the free energy of forma-
tion of the proposed H
x
CuO
y
species, the lack of which had been criticised in reviews of earlier
papers from this group. Hultquist et al. (2009) chose to perform simulations for the copper atom-
hydroxylradicalsystem,forwhichtheyestimatedaΔG
f
value of –548 kJ/mol. This value should
be compared with values of –200 kJ/mol (Wagman et al. 1982, cited by Apted at al. 2009) and the
value of –228 kJ/mol for CuOH
ADS
(Protopopoff and Marcus 2005). Using the value estimated using
molecular dynamic simulations gives an overall free energy change for the reaction
Cu + H
2
O→CuOH+½H
2
(2-11)
of–311kJ/mol,comparedwithvaluesof+37kJ/moland+9kJ/molbasedontheΔG
f
values for
CuOH of Wagman et al. (1982) and Protopopoff and Marcus (2005), respectively. Based on the value
estimated for CuOH by Hultquist et al. (2009), the equilibrium H
2
pressure for Reaction (2-11) is
>10
100
bar at 25°C.
The experimental measurements were similar to those reported by Szakálos et al. (2007) and carried
out in a similar apparatus, except with the addition of a pressure gauge and a facility to sample the
gas phase for analysis using a mass spectrometer. Few details were given of the results of this test,
although later in the text the authors refer to an equilibrium H
2
pressure of 0.5 mbar at 45°C. This
value is clearly inconsistent with that calculated from the free energy change for Reaction (2-11).
Hultquist et al. (2009) predict the extent of corrosion of a copper canister based on a linear extrapo-
lation of a log-log plot of the time dependence of the depth of corrosion based on measurements for
four different exposure times. The basis for each of these four points is unclear, but from what is
stated in the text and from estimates taken directly from Figure 2-9, it appears that:
• ThedatumfortheshortestexposureperiodisbasedonthedealloyingofaCu-Aualloyinwater
for a period of 20 hrs. Based on a measurement directly from Figure 2-9, the depth of de-alloying
appears to have been ~0.3 nm, giving a mean "corrosion" rate of 0.1 µm/yr. Quite what relevance
the de-alloying of a Cu-Au alloy has for the corrosion of a copper canister is unclear.
• Thesourceofthedatumatatimeof~0.1yrisunknown.Hultquistetal.(2009)refertoa
"Ref. [24]" but this relates to an investigation of the condition of the 17
th
century Swedish war-
ship Vasa and a careful reading of that article makes no mention of copper artifacts. Regardless,
the depth of corrosion from the figure is ~0.02 µm in a period of 7 weeks giving a mean corro-
sion rate of 0.15 µm/yr.
• Thedatumatatimeof15yrscomesfromthestudyofHultquistetal.(2008).Thetextrefersto
a maximum depth of 35 µm, although direct measurement from the figure suggests a value of
14 µm. Regardless, the mean corrosion rate is of the order of 1–2 µm/yr.
• Thefinaldatapointsatatimeof333yrsareestimatedfromobservationsofcoppercoinsfrom
the warship Vasa. Measurements from the figure suggest a corrosion depth of 1.1 mm, giving a
mean corrosion rate of 3.3 µm/yr. A more-detailed discussion of the relevance of these artifacts
is given below.
Based on these four data points, Hultquist et al. (2009) then linearly extrapolated the log-log plot and
estimated that a copper canister would corrode more than 1 m in 100,000 yrs. (In fact, extrapolation
of the data suggests a depth of ~3 m, equivalent to a mean corrosion rate of 30 µm/yr). A feature
of this extrapolation of a log-log plot, which Hultquist and co-workers acknowledged, is that the
corrosion rate is predicted to increase with time. Hultquist et al. (2009) claimed that this was due to
the uptake of H by the copper, and cited evidence for a similar effect on steels and copper. The refer-
ence cited for the effect of H on steels (Yang et al. 1998, the reference is mis-cited as Qiao and Luo
1998 by Hultquist et al. 2009) actually refers to the effects of H on the localised corrosion properties
of passive films on Type 310 stainless steel. Whilst evidence for an increase in the rate of general
corrosion of C-steel due to absorbed H has also been reported in the literature (Gu et al. 1999), the

TR-10-69 29
effect seems to require severe charging conditions (24 h cathodic charging at a current density of
3 mA·cm
–2
in a 1 mol·dm
–3
H
2
SO
4
solution containing 250 µg·g
–1
As
2
O
3
). The reference cited for the
effect of hydrogen on copper corrosion refers to a Sandia National Labs news bulletin that describes
experiments with implanted D in thin vapour deposited Cu layers on Si wafers (Sandia 2000).
The most important set of data in Figure 2-9 are those from the Vasa since they represent the longest
exposure time of any of the points on the plot. Hultquist et al. (2009) claimed that, as with the other
data on the figure, the artifacts from the Vasa had been exposed to anoxic water. This was based on
the report by Sandström et al. (2002) on the degradation of the wooden structure of the ship after
exposure to air. Figure 2-10 shows an image of two of the coins on which Hultquist et al. (2009)
based their estimate of the corrosion rate. One coin is clearly more-severely corroded than the
other, which Hultquist et al. (2009) claimed was due to limited access of water to the less-severely
corroded coin. It is highly unlikely that the rate of water access would vary for different coins in
saturated media. A far more likely explanation is that the corrosion rate was limited by the supply
of sulphide. Sandström et al. (2002) refer to an earlier measurement of a sulphide concentration of
7 mg/L in the region of the Vasa. Sulphide is present because the anoxic conditions were created
by microbial activity in the vicinity of the wreck. It is the oxidation of that sulphide and elemental
sulphur that is now causing deterioration of the wooden components of the ship in the Vasa museum.
In summary, the scientific basis for the data in Figure 2-9 and the conclusions drawn from
extrapolation of the corrosion damage must be considered of dubious validity.
Figure 2-9.
Log-log plot of corrosion depth against time for copper-containing alloys in assumed anoxic
conditions used by Hultquist et al. (2009) to predict the depth of corrosion on a copper canister (Figure 4
from Hultquist et al. 2009).
Figure 2-10.
Image of copper coins taken from the wreck of the Vasa (Figure 3 from Hultquist et al. 2009).
30 TR-10-69
2.1.13 Korzhavyi and Johansson, SKB TR-10-30, 2010
Korzhavyi and Johansson (2010) used quantum mechanical techniques to investigate the structural
and energetic stability of various compounds of Cu
+
, Cu
2+
, O
2–
, H
–
and H
+
. The aim of the studies
was to determine, using density functional theory and linear response theory, whether the H
x
CuO
y
species proposed by Szakálos et al. (2007) was stable.
The authors first validated their approach by calculating enthalpies and free energies of formation of
compounds that have been well studied and for which experimentally measured values are available.
The compounds considered (and their structures) were Cu
2
O (cuprite structure), CuO (tenorite), and
CuH (wurtzite and sphalerite structures). All species were found to be structurally stable and the
calculated enthalpies and free energies of formation close to the measured values.
Having thus validated their computational approach, Korzhavyi and Johansson (2010) then went
on to assess the stability of other Cu, O, and H-containing compounds that corresponded to the pro-
posed H
x
CuO
y
species. Various structural forms of Cu
4
H
2
O and CuH
2
were found to be dynamically
unstable. Different structural forms of CuOH were also investigated and a free energy of formation
of –158 kJ/mol to –161 kJ/mol estimated. These values contrast with the value of –584 kJ/mol pro-
posed by Hultquist et al. (2009) based on molecular dynamics simulations of copper atoms and the
hydroxyl radical, but are of the same order as the values of –200 kJ/mol and –228 kJ/mol reported by
Wagman et al. (1982) and Protopopoff and Marcus (2005).
TakingaΔG
f
value of –159 kJ/mol for CuOH produces a free energy change for reaction (2-11) of
+78 kJ/mol. Furthermore, CuOH is metastable with respect to Cu
2
O since the free energy change for
the reaction
2CuOH = Cu
2
O + H
2
O (2-12)
is –66 kJ/mol.
Thus, the quantum mechanical simulations of Korzhavyi and Johansson (2010) demonstrate that
CuOH could not be the stable solid phase responsible for the corrosion of copper by water.
2.1.14 Bojinov et al. Corros. Sci. 2010
Bojinov et al. (2010) carried out a series of careful electrochemical measurements on copper in
deaerated deionised water in an attempt to identify possible reactions that could account for the
generation of H
2
from this system. Very low concentrations of dissolved Cu
2+
were observed in solu-
tion (of the order of 20 nmol/L), thought to be generated by the dissolution of the air-formed oxide
produced during preparation of the electrode or by dissolution of oxide formed by the consumption
of trace levels of residual O
2
in solution. The similarity of the open-circuit potential of a copper
electrode and that of a Cu
2+
ion-selective electrode suggested that the potential was determined by a
redox reaction involving the dissolved Cu
2+
. The presence of a Cu
2
O film was confirmed during the
early stages of the exposure (after 2-3 hours) by cathodic stripping voltammetry. However, no such
film was present after a total exposure period of 330 hrs, suggesting that further corrosion of the
electrode had not occurred following the consumption of the residual O
2
. There was also evidence
for adsorbed surface species from electrochemical impedance spectroscopy measurements.
TR-10-69 31
The observations were interpreted in terms of an adsorbed CuOH
ads
species responsible for the
reduction of water by a reaction such as
Cu*(O)H
ads
+ H
2
O + e
–
= Cu*(O)
ads
+ H
2
+ OH
–
(2-13)
where Cu* indicates a surface copper atom.
Two possible routes for the formation of CuOH
ads
(or Cu*(O)H
ads
) were considered, one involving
the oxidation of Cu
H
2
O
ads
= OH
ads
+ H
+
+ e
–
(2-14a)
OH
ads
+ Cu = CuOH
ads
(2-14b)
and the other involving the reduction of dissolved Cu
2+
Cu
2+
+ OH
–
+ e
–
= CuOH
ads
(2-15)
Reaction (2-14) can be written more simply as
Cu + H
2
O
ads
= CuOH
ads
+ H
+
+ e
–
(2-16)
where the fact that this reaction involves oxidation of the copper is clear.
Fitting of this model to their electrochemical measurements allowed the rate constants for the vari-
ous reactions to be determined, from which it was apparent that the predominant source of CuOH
ads
was the reduction of dissolved Cu
2+
(reaction (2-15)). It was concluded, therefore, that H
2
could be
produced by reaction (2-13) without the requirement to oxidise copper.
2.2 Summary of key points from the critical review of
the literature
There are a number of key points arising from the various studies reviewed above. These key issues
are summarised here in bullet form, with a more-detailed discussion provided in the foregoing
sections:
• Hultquistandco-workershaveprovidedevidencefortheevolutionofH
2
during the corrosion
of copper in pure water based on: (i) the difference in corrosion rate and visual appearance of
samples in Pd- and Pt-sealed vessels, (ii) ion-pump measurements of the flux of H
2
through a
Pd membrane, (iii) pressure changes due to the consumption of O
2
and the production of H
2
.
• Gråsjöetal.(1995)didnotobserveH
2
production from the corrosion of copper in water,
although the experiment may not have been allowed to run long enough for anoxic conditions
to be established.
• NeitherSimpsonandSchenk(1987)norEriksenetal.(1988,1989)observedH
2
production
from the corrosion of copper in aqueous chloride solutions or pure water, respectively, using
gas chromatography.
• Möller(1995)didnotseeanyvisualdifferenceintheappearanceofCustripsinPd-and
Pt-sealed glass vessels and corrosion appears to have ceased half-way through the 2-year
experiment.
• ThepotentialofCuwireduringsupposedH
2
generation is 150 mV more positive than the
equilibrium potential for the H
2
O/H
2
rection for a H
2
pressure of 1 atm (Seo et al. 1987).
• TheproductionofH
2
has been associated with the formation of a previously unknown phase
H
x
CuO
y
(Hultquist et al. 2008, 2009, Szakálos et al. 2007).
• TheequilibriumH
2
pressure for the Cu/ H
x
CuO
y
system is believed to be ~1 mbar (101 Pa) at
73°C (Szakálos et al. 2007).
32 TR-10-69
• Iftheion-pumpcurrentdescribedintheworkofSzakálosetal.(2007)isameasureofthefluxof
H
2
as the authors claim, then their results indicate that the rate of corrosion increases monotoni-
cally upon exposure to O
2
-free water.
• Hultquistandco-workerspointtothepresenceofHincorrosionproductsoncopperasevidence
for the reduction of H
2
O, but passive films, including those on copper, are hydrated to some
degree.
• ThecorrosionofcopperinwaterwillcontinueprovidedH
2
is removed from the interface and the
pressure remains below the equilibrium pressure (Hultquist 1986, Hultquist et al. 1989, Szakálos
et al. 2007).
• Oxygendoesnotsupportthecorrosionofcopperinwaterbutis,instead,consumedbyreaction
with H atoms produced by the reduction of H
2
O (Gråsjö et al. 1995, Hultquist 1986, Hultquist
et al. 2008, Seo et al. 1987, Szakálos et al. 2007).
• ThereductionofH
2
O is capable of supporting the oxidation of copper to the Cu(I) and Cu(II)
oxidation states (Hultquist et al. 1989).
• BasedontheanalysisbytheSSMBRITEgroupofthepaperbySzakálosetal.(2007),the
corrosion rate of copper in the "pressure-gauge" experiment is approximately 40 times lower
than that in the ion-pump experiment despite being done at a higher temperature.
• Quantummechanicalcalculationsindicatethattherearenostable(withrespecttoCu
2
O)
compounds that would correspond to the proposed H
x
CuO
y
species proposed by Hultquist and
co-workers (Korzhavyi and Johansson 2010).
• Bojinovetal.(2010)suggestthatH
2
could be formed without the oxidation of copper due to
reactions between an adsorbed CuOH
ads
species and H
2
O. The CuOH
ads
species is formed from
the reduction of dissolved Cu
2+
that results from the dissolution of a pre-formed Cu
2
O layer or
from the consumption of residual O
2
.
• Hultquistandco-workershavereportedH
2
generation from initially aerated experiments
(e.g. Hultquist (1986)) and from experiments in which the water used had been extensively
deaerated (e.g. Hultquist et al. (2009)). Thus, pre-oxidation of the surface does not seem to
affect H
2
generation from copper.
• NeitherHultquistandco-workersnorotherresearchershavereportedthegenerationofH
2
when
copper is exposed to solutions other than pure water, e.g. saline solutions representative of
bentonite pore water or deep ground waters.
TR-10-69 33
3 Supplementary analyses
In this Section, we expand on some of the issues raised from the critical review of the literature in
Section 2 and provide further analyses and discussion in an attempt to resolve these issues.
3.1 Formation and properties of adsorbed Cu-OH species
Hultquist and co-workers have proposed the formation of an unidentified stable solid phase to
account for the evolution of H
2
from the corrosion of Cu in pure water. Based on SIMS analysis of
corrosion products from aqueous- and gas-phase experiments, this solid has been identified by the
generic composition H
x
CuO
y
. The thermodynamic properties of this species have been inferred from
an apparent equilibrium partial pressure of H
2
observed by Szakálos et al. (2007). Although, presum-
ably, x and y could be non-integral, the corresponding compounds for x = y = 1 and x = y =2 are
CuOH and Cu(OH)
2
. The latter, of course, exists as a Cu(II) solid and its thermodynamic properties
are known (Pourbaix 1974), but it is only stable at potentials approximately 700 mV more positive than
the H
2
O/H
2
equilibrium (Figure 1-1(b)). The species CuOH is unknown as a stable solid (although
Wagman et al. (1982) do provide a single value for the free energy of formation), but is thought to
exist as a surface intermediate species formed during the oxidation of copper in electrochemical
experiments (King and Kolar 2000).
Figure 3-1 shows the results of cyclic voltammetric experiments in deaerated 0.1 mol·dm
–3
NaClO
4
solution at pH 7 and pH 10 (King et al. 1995a). The anodic peaks A
1
and A
2
on the forward potential
scan are believed to be due to the formation of sub-monolayer quantities of Cu(OH)
ADS
or Cu
2
O,
formed by the reactions (Burstein and Newman 1981, Elsner et al. 1988, King and Kolar 2000):
Cu + H
2
O→Cu(OH)
ADS
+ H
+
+ e
–
(3-1a)
or
2Cu + H
2
O→Cu
2
O + 2H
+
+ 2e
–
(3-1b)
Formation of Cu
2
O by the loss of water from Cu(OH)
ADS
2Cu(OH)
ADS
→Cu
2
O + H
2
O (3-2)
is also possible but, being a chemical reaction, would not account for the peaks seen in Figure 3-1.
Peak A
1
is more prominent at the higher pH (Figure 3-1(a)), suggesting that this species could be
due to the formation of Cu(OH)
ADS
. Peak C
1
is due to the reduction of Cu
2
O (i.e. the reverse of
Reaction 3-1(b)), as identified from the peak potential. Peaks A
1
and A
2
account for less than one
monolayer of adsorbed species, based on the monolayer charge densities given in the literature (see
King and Kolar 2000). A monolayer or more of Cu
2
O is only formed if the potential is scanned into
region A
3
.
An important feature of peak A
1
is that it is formed at a potential more negative than the equilibrium
potential for Reaction (3-1(b)). At pH 10, the equilibrium potential for the formation of Cu
2
O is
–0.36 V
SCE
(Pourbaix 1974), compared with a peak potential for A
1
of –0.65 V
SCE
.
In chloride solutions, there appears to be competition between OH
–
and Cl
–
for surface sites
(King and Kolar 2000). Chloride ions are known to form adsorbed CuCl
ADS
species at potentials up
to 700 mV more-negative than the equilibrium potential (Elsner et al. 1988). Figure 3-2 shows the
voltammetric behaviour of Cu in deaerated 1 mol·dm
–3
NaCl solution at pH 10 (King et al. 1995b).
In this case, peak A
2
is believed to be the formation of sub-monolayer CuCl
ADS
at a potential 700 mV
below the equilibrium potential (–0.105 V
SCE
, Moreau 1981). Peak C
1
is again due to the reduction
of Cu
2
O and is observed if the potential is scanned anodically into region A
3
. (In this case, peak
A
1
is believed to be due to the oxidation of adsorbed or absorbed H formed during the cathodic
pre-cleaning at –1.3 V
SCE
(Elsner et al. 1988, King and Kolar 2000)). In 1 mol·dm
–3
Cl
–
solution,
therefore, Cl
–
wins the competition for surface sites even in pH 10 solution.

34 TR-10-69
Figure 3-1.
Cyclic voltammetric studies of the formation of surface films on copper in deaerated
0.1 mol·dm
–3
NaClO
4
solution at room temperature (King et al. 1995a).

TR-10-69 35
These sub-monolayer adsorbed species are believed to catalyse certain reactions on the copper
surface (King et al. 1995a, b). Figure 3-3 shows a series of potential scans in oxygenated unbuffered
0.1 mol·dm
–3
NaClO
4
solution. Scans were started with a pre-cleaned surface at a potential of
–1.2 V
SCE
and scanned to more-positive potentials in each successive scan. Up to an upper potential
limit of –0.55 V
SCE
(curve (c)), the rate of O
2
reduction was the same on both the forward and
backward scans. With increasing potential limit, however, a peak appeared on the forward scan
(see curve (g)), with a higher rate of O
2
reduction on the forward (anodic-going) scan. This enhanced
current is lost, however, on the reverse (cathodic-going) scan.
The potential-dependence of the catalysis of the O
2
reduction reaction in Figure 3-3 has been associ-
ated with the formation of surface films shown in Figure 3-1. Based on measurements of the interfa-
cial pH during O
2
reduction (King et al. 1995a), it was shown that the pH at the electrode surface is
~pH 10 in the limiting current region during the reduction of O
2
in oxygenated solution. Therefore, it
is assumed that the surface films observed in Figure 3-1(a) are those present during O
2
reduction on
Cu in unbuffered solution. That being so, it can be seen that catalysis of the O
2
reduction reaction is
associated with the presence of sub-monolayer Cu(OH)
ADS
or sub-monolayer Cu
2
O. Formation of a
complete monolayer of Cu
2
O, as happens for an upper potential limit of –0.1 V
SCE
results in a loss of
catalytic activity (curve (g), Figure 3-3).
The question is whether these same catalytic sub-monolayer surface films also catalyse H
2
evolution.
Inspection of Figures 3-1 and 3-2 suggests that this is not the case. In each figure, the current in the
region of H
2
O reduction (i.e. at potentials more negative than approximately –1.0 V
SCE
) is lower on
the reverse scan (when catalytic sub-monolayer surface films are present) than on the forward scan
on clean Cu surfaces. Therefore, even if Cu(OH)
ADS
is present on the surface, it would not support
enhanced H
2
evolution.
Figure 3-2.
Cyclic voltammograms on copper in deaerated 1 mol·dm
–3
NaCl solution at pH 10 at room
temperature (King et al. 1995b).
36 TR-10-69
Figure 3-3.
Series of cyclic voltammograms for a rotating copper disc electrode in oxygenated unbuffered
0.1 mol·dm
–3
NaClO
4
solution at room temperature. The scans were performed from a potential of –1.2 V
SCE
to ever increasing upper potential limits in successive scans (a) to (g). For clarity, the curves have been
offset from each other by increments of 500 µA. Potential scan rate 10 mV/s (King et al. 1995a).
As noted above, the sub-monolayer Cu(OH)
ADS
(or Cu
2
O) and CuCl
ADS
form at potentials more-
negative than the equilibrium for the bulk Cu
2
O and CuCl phases, respectively. The potentials of
peaks A
1
and/or A
2
can be used to derive the free energy of formation of the respective species
(Protopopoff and Marcus 2005). Based on the in situ scanning tunneling microscopy and electro-
chemical experimental evidence of Maurice et al. (2000), who observed a reversible potential for
Cu(OH)
ADS
formation of –0.916 V
SCE
at pH 13, Protopopoff and Marcus (2005) estimated a free
energy of formation of –228 kJ/mol. This value was then used to construct a modified Pourbaix
diagram for the Cu-H
2
O system that included an adsorbed Cu(OH)
ADS
species superimposed on
top of the conventional E-pH diagram for this system (Figure 3-4).
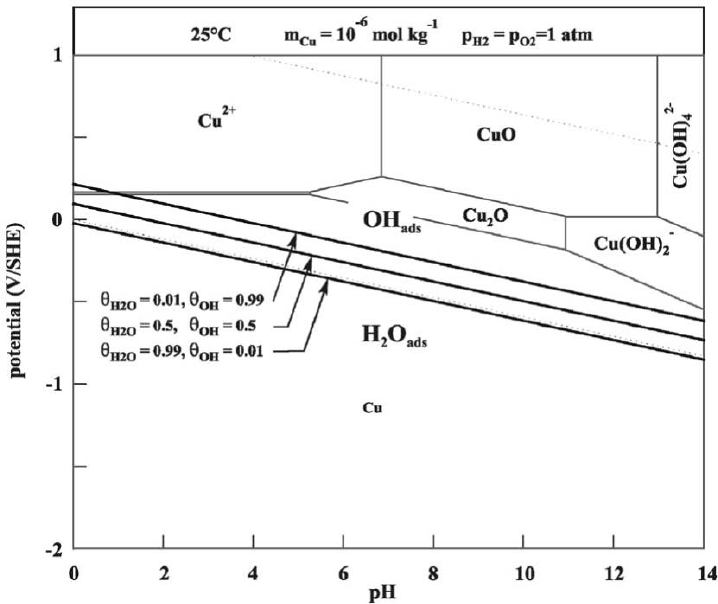
TR-10-69 37
Figure 3-4 shows a stability region for the species Cu(OH)
ADS
that extends below that for Cu
2
O.
This figure is similar to that shown by Szakálos et al. (2007). The series of three parallel lines on
the modified Pourbaix diagram represent the equilibrium for the reaction (in the notation used by
Protopopoff and Marcus (2005))
H
2
O
ads
(Cu) = OH
ads
(Cu) + H
+
+ e
–
(3-3)
as a function of surface coverage by the two adsorbed species. The stability field for the adsorbed
hydroxide species only extends below the H
2
O/H
2
equilibrium line (for a H
2
partial pressure of
1 atm.) for surface coverages of less than approximately 0.03 monolayers.
Although this analysis extends the region in which copper may be oxidised, in this case due to the
electroadsorption of OH
–
species, the question is what impact this has on the corrosion of copper in
pure water and the possible generation of H
2
. The electroadsorption of OH
–
has only been observed
on cathodically cleaned copper surfaces and only in sub-monolayer coverages. The electrochemical
evidence presented above suggests that if the surface coverage is allowed to approach the monolayer
level then the adsorbed hydroxide species converts to Cu
2
O via Reaction (3-2). The surfaces used
by Hultquist and co-workers were mechanically polished but not subsequently electrochemically
cleaned. These surfaces would, therefore, have been covered by an air-formed oxide and the forma-
tion of sub-monolayer CuOH
ADS
species as discussed above would not have occurred. Furthermore,
in order to sustain the electroadsorption process and the possible coupled evolution of H
2
, the surface
coverage of adsorbed OH
–
would have to be maintained at the sub-monolayer level because the equi-
librium line shifts to more-positive potentials with increasing surface coverage. Without a process
for limiting the surface coverage of Cu(OH)
ADS
, the possible evolution of H
2
would be short-lived.
Figure 3-4.
Pourbaix diagram for the Cu–H
2
O system at 25°C including the regions of stability of
Cu(OH)
ADS
for various surface coverages θ (Protopopoff and Marcus 2005).
38 TR-10-69
3.2 Corrosion of copper in anoxic concentrated
chloride solutions
Copper is thermodynamically unstable in water at low pH in the presence of high concentrations
of chloride ions (Figure 1-2). Although high Cl
–
concentrations (greater than, say, 1 mol·dm
–3
) are
possible in deep groundwaters, especially in Finland (King et al. 2001), it is unlikely that acidic
conditions will develop. Not only will the near-field pH be buffered by the presence of bentonite
and calcite, but the locally acidic conditions in occluded regions are not expected to occur for
copper. Local acidification is unlikely because Cl
–
strongly stabilises dissolved Cu(I), which does
not hydrolyse strongly in any case, and because Cu(II), which does strongly hydrolyse, will not
be formed either by direct dissolution at the corrosion potentials expected in the repository or in
occluded regions because of the absence of O
2
to oxidise Cu(I).
Although Cu is predicted to be thermodynamically stable in O
2
-free Cl
–
solutions at neutral pH,
Posiva have conducted a limited experimental programme to establish if corrosion is possible under
these conditions (Bojinov and Mäkelä 2003, Bojinov et al. 2004). Copper in the form of mass-loss
coupons and as an electrical resistance probe was exposed to 1 mol·dm
–3
NaCl solution at room
temperature or 80°C for periods up to 30 days. The solution was deaerated after placing the coupons
in the test vessel, but prior to the start of the experiment, by bubbling O
2
-free N
2
through the solution
for 4–6 hrs. This technique typically leaves a few ppb residual O
2
(Butler et al. 1994, King et al.
2001), although Bojinov and Mäkelä (2003) presented evidence suggesting a dissolved [O
2
] of
0.001–0.01 ppb. The corrosion rate was measured in situ using the electrical resistance probe, with a
time-averaged value also obtained from the mass-loss and solution analyses. The redox potential of
the solution was also measured using a Pt electrode.
The corrosion rate was observed to decrease with time and then stop after 60–80 hrs exposure (Bojinov
and Mäkelä 2003, Bojinov et al. 2004). The authors attributed this behaviour to the consumption of
residual O
2
followed by the cessation of corrosion once anoxic conditions had been established. The
solution pH was found to increase from an initial value of pH 6.4 to a final value of pH 9.5–9.9, the
increase being attributed to the OH
–
ions produced by the reduction of the residual O
2
. No H
2
was
detected in the headspace of the autoclave following a 6-day exposure, consistent with the absence of
H
2
O reduction. No further H
2
measurements were made because of failure of the gas sampling system.
The corrosion potential (E
CORR
) was also measured during the tests. An apparent steady-state
E
CORR
was attained after ~15 days exposure, with a value of –0.33 V
SCE
at room temperature and
–0.43 V
SCE
at 80°C. The value at room temperature is more-positive than that reported by King et al.
(1995c) for a copper rotating-disc electrode in nominally deaerated 1 mol·dm
–3
NaCl, but this is
consistent with the negative shift in E
CORR
with increasing rate of mass transport as a consequence
of the mass-transport limitation of the anodic reaction (King et al. 1995c).
More interestingly, however, the establishment of a steady-state E
CORR
was found to correspond to
the time at which corrosion stopped. Furthermore, the value of the steady-state E
CORR
was at the
boundary of the region of immunity on the Pourbaix diagram, as defined by the Cu/CuCl
2
–
equilib-
rium line for a dissolved copper concentration of 10
–5
–10
–6
mol·dm
–3
(Figure 1-2).
In conclusion, therefore, there is no evidence for the corrosion of copper in O
2
-free 1 mol·dm
–3
NaCl
solution at room temperature or 80°C. Corrosion, directly measured in situ using an electrical resist-
ance technique, ceased once the residual O
2
had been consumed. This observation is supported by
the absence of H
2
in the one gas sample analysed. Furthermore, the cessation of corrosion occurred
at a potential at which the conventional Cu-H
2
O-Cl
–
E-pH diagram predicts the immunity of copper.
3.3 Corrosion of copper in compacted bentonite
King et al. (1997) studied the corrosion of copper in compacted bentonite under simulated repository
conditions. The aim of the study was to determine the environmental conditions under which Cu(I) or,
alternatively Cu(II), was the major corrosion product. To this end, the bentonite clay was compacted
with, and immersed in, synthetic ground waters with Cl
–
concentrations of between 0.17 mol/L
and 2.5 mol/L, with Cu(I) expected to be progressively stabilised with increasing [Cl
–
]. The redox
conditions in the sealed glass vials in which the tests were conducted were also varied, with initial
TR-10-69 39
atmospheres of either 0.23 vol.% O
2
or air (20.9 vol.% O
2
). In some cases, Fe filings were included
to the dry clay before compaction in order to scavenge O
2
during the test and produce anoxic
conditions. Clearly, the formation of Cu(II) was expected to be favoured by increased [O
2
] and the
absence of Fe filings. All tests were conducted at a temperature of 95°C for periods of between
10 and 184 days with an initial gas pressure of 1 atm. Based on previous work, the predominant
copper oxidation state was indicated by the shape of the copper diffusion profile in the clay and the
nature of the precipitated corrosion product. Of more interest for the current discussion, however, is
the fact that the atmosphere inside the sealed glass vial was sampled at the end of the test and the gas
composition determined, including the presence of H
2
.
Detailed results from the various tests, as well as from an aerated blank test that contained all of the
test components with the exception of the copper coupon, are reported by King et al. (1997). The
observations relevant to the current subject include:
• Intheoneblanktestperformed,theinitialairatmospherewasfoundtohavechangedtoacomposi-
tion of 0.043 vol.% H
2
, 3.51 vol.% O
2
, 1.17 vol.% Ar, 5.29 vol.% CO
2
, balance N
2
at the end of the
test. The Ar content is similar to that in air, suggesting a final pressure of close to 1 atm. and that the
results are reliable. The decrease in O
2
and increase in CO
2
were attributed to microbial activity. Of
interest is the generation of H
2
in the complete absence of corrodible metals, including copper.
• Ofthe27experimentsforwhichgasanalyseswereperformed,H
2
was observed at concentrations
above the detection limit (0.001 vol.%) in 25 of the tests.
• Ofthe25experimentsinwhichmeasureableH
2
was detected, 11 tests gave H
2
concentrations
below that found in the blank test. In these cases, the unknown reaction(s) responsible for H
2
generation in the blank test is(are) considered responsible for the observed H
2
.
• Ofthe14remainingtests,eight(withobservedH
2
concentrations of 0.28–3.3 vol.%) contained
Fe filings, with the source of H
2
considered to be the corrosion of Fe.
• Allofthe6remainingexperimentsinwhichH
2
was observed at concentrations higher than in
the blank test were performed under initially aerated conditions with between 5.4 vol.% and
17.5 vol.% O
2
remaining at the end of the test. The concentration of H
2
observed varied between
0.057 vol.% and 0.43 vol.%. The observed H
2
concentrations may be within the (unknown)
scatter of the blank experiment.
The source of the H
2
in tests without Fe filings is unknown. What is clear, however, is that H
2
in
excess of that observed in the blank test (without a copper coupon) was only found under aerobic
conditions, quite unlike those that Hultquist and co-workers suggest are required for the oxidation of
copper by water.
3.4 Role of dissolved oxygen
In a number of their publications, Hultquist and co-workers claim that residual O
2
in the test cell
is consumed by reaction with atomic H produced by the reduction of H
2
O (Gråsjö et al. 1995,
Hultquist 1986, Hultquist et al. 1989, 2008, Seo et al. 1987, Szakálos et al. 2007). This claim is
based primarily on analogy with the behaviour in gas-phase systems that have been studied by
Hultquist and some of his colleagues. If correct, this claim would not only mean that H
2
O is an
oxidant for copper but also that H
2
O is preferentially reduced compared with O
2
. Furthermore, it
would seem to indicate that O
2
itself is not directly an oxidant for copper.
This somewhat unconventional claim warrants further discussion, not least because it directly affects
the common understanding of the corrosion behaviour of metals in aqueous systems.
Many researchers have investigated the corrosion behaviour of copper and copper alloys in
O
2
-containing environments. For the conditions expected in a deep geological repository, King and
co-workers have studied the corrosion of copper in O
2
-containing chloride solutions over a broad
range of conditions, including the effects of temperature, Cl
–
concentration, O
2
partial pressure, pH,
the rate of mass transport, and the presence of various ions and complexants, including SO
4
2–
and
NH
4
+
(King et al. 2001). Based on the results of electrochemical studies and of experiments in com-
pacted bentonite, King and Kolar (2000) developed a reaction scheme that described the corrosion
behaviour of copper canisters in the repository environment (Figure 3-5). There are two roles for

40 TR-10-69
Figure 3-5.
Reaction scheme for the corrosion of copper canisters in a deep geologic repository
(King and Kolar 2000).
dissolved O
2
within this reaction scheme, namely (i) the interfacial cathodic reduction of O
2
to OH
–
and (ii) the homogeneous oxidation of Cu(I) to Cu(II) by dissolved O
2
. Neither of these processes
is considered in reaction schemes proposed by Hultquist and co-workers. Evidence for the latter
reaction is abundant in the literature (e.g. Sharma and Millero 1988) and is not discussed further
here. Evidence for the important role played by the interfacial reduction of O
2
is discussed below.
The corrosion of copper in O
2
-containing Cl
–
solutions involves the anodic dissolution of copper as
cuprous-chloro complex ions, such as CuCl
2
–
, coupled to the cathodic reduction of O
2
. The anodic
dissolution of copper has been extensively studied (Kear et al. 2004). From their own studies (King
and Kolar 2000, King et al. 1995c), King and co-workers were able to identify (i) the mechanism
of the two-step interfacial process, (ii) the nature of the mass-transport rate determining step, and
(iii) values for the diffusivity of CuCl
2
–
and of various rate constants. King and co-workers also
studied the cathodic reduction of O
2
on copper (King et al. 1995a, b) and deduced the mechanism
and the values of interfacial rate constants and other Tafel characteristics. This mechanistic informa-
tion was then used to develop a steady-state mixed-potential model from which, by using the values
of the independently measured anodic and cathodic parameters, predictions of the E
CORR
were made
for a range of environmental conditions (King et al. 1995c).
The validity of the proposed reaction mechanism and the derived mixed-potential model was tested
by comparing predicted E
CORR
values with those measured experimentally (King et al. 1995c).
Excellent agreement (within a few mV) was obtained between experiment and model for a range of
fiveordersofmagnitudevariationinthesteady-statemass-transfercoefficient(D/δ,whereDisthe
diffusivityandδisthediffusionlayerthickness),variationintheconcentrationofdissolvedO
2
by
a factor of 100–1,000, and for a 10-fold variation in Cl
–
concentration. As an example, Figure 3-6
shows a comparison of the measured and predicted E
CORR
for a copper electrode covered by a 1-mm-
thicklayerofcompactedbentonite(D/δ=10
–6
cm·s
–1
) in O
2
-containing 1 mol·dm
–3
NaCl solution as
the purge gas is changed from air to 2 vol.% O
2
/N
2
, to 0.2% O
2
/N
2
, and eventually to pure N
2
(King
et al. 1995c). Measured and predicted E
CORR
values agree within 15 mV.
2
-
J
O2
2
+O
2
k
5
k
7
k
-7
PPT
k
C
2+
2
2
-
ADS
k
bf
k
bb
k
-2
k
2
2+
k
af
k
ab
k
D
k
3
k
-3
2+
2
-
k
1
2 2
k
6
J
CuCl2-
J
Cu(II)
k
-4
k
4
ADS
AQ
Aerobic
microbes
k
9
2
k
8
k
-7
k
7
PPT
TR-10-69 41
Figure 3-6.
Comparison of measured time-dependent (sold curve) and predicted steady-state (horizontal
dashed lines) corrosion potentials for copper in contact with a 1-mm-thick layer of compacted bentonite
immersed in O
2
-containing 1 mol·dm
–3
NaCl solution at room temperature (King et al. 1995c). The solution
was initially aerated and then the purge gas was changed to the indicated composition at the times
indicated by the respective arrows.
This independent validation of measured and predicted E
CORR
values is strong evidence in support
of the role of O
2
as the oxidant for the corrosion of copper in O
2
-containing solutions. Furthermore,
this evidence calls into question the claim by Hultquist and co-workers that O
2
is consumed by the
products of H
2
O reduction.
3.5 Corrosion potential considerations
The discussion in the previous section centred on the measurement and prediction of E
CORR
. The
value of E
CORR
can be used to provide valuable insight into the corrosion mechanism and can be
used to predict what reactions are occurring or might occur, as discussed in the examples below.
By definition, E
CORR
must lie between the equilibrium potentials for the anodic and cathodic reac-
tions. If we assume that the proposed mechanism is correct, then the maximum value of E
CORR
will
be determined by the equilibrium potential for the reaction
H
2
O + e
–
→½H
2
+ OH
–
(3-4)
where the arrow should strictly be reversible if the process is at equilibrium. Based on the reported
equilibrium pressure of 1 mbar at 73°C (Szakálos et al. 2007), the BRITE group estimated a redox
potential of –0.343 V
SHE
(or –0.58V
SCE
assuming the same conversion factor as at 25°C) (Apted at
al. 2009). This maximum value of E
CORR
is 80 mV more negative than the room-temperature value
reported by Seo et al. (1987) and several hundred mV more negative than the experimental values of
King et al. (1995c) at room temperature and 150 mV more negative than the value of Bojinov et al.
(2004) at 80°C.
42 TR-10-69
Hultquist et al. (1989) even suggested that H
2
O could oxidise copper to the Cu(II) state. The authors
proposed the following reaction sequence:
Cu + H
2
O→CuOH+H (3-5a)
followed by
2CuOH→Cu
2
O + H
2
O (3-5b)
or
CuOH→CuO+H (3-5c)
Reaction 3-5(c) represents the oxidation of Cu(I) to Cu(II) with the formation of H atoms. If correct,
based on the Pourbaix diagram in Figure 1-1(a), this would suggest that hydrogen is being evolved at
a potential of +0.014 V
SCE
.
Based on the limited available information on the E
CORR
of copper in deaerated systems, therefore, it
seems unlikely that the potential is sufficiently negative to allow for the reduction of H
2
O.
3.6 Implications for corrosion of a copper canister in a deep
geologic repository
In attempting to make predictions of the extent of canister corrosion, Hultquist et al. (2009) fail to
properly take into account the repository system and the repository environment. Apart from the
questionable basis for the corrosion rates that they use in their extrapolation, Hultquist et al. (2009)
ignore one of their own basic findings from their studies. If we accept, for the sake of argument, that
their proposed mechanism is correct then, by their own evidence, the extent of canister corrosion due
to the reduction of water would be minimal.
A basic tenet of the proposed mechanism is that there exists an equilibrium partial pressure of H
2
below which copper will corrode with H
2
O as an oxidant. Equally, as they repeatedly emphasise
(Hultquist 1986, Hultquist et al. 1989, 2009, Szakálos et al. 2007), corrosion will cease once that H
2
partial pressure is reached. The proposed equilibrium pressure is ~1 mbar at 73°C (Szakálos et al.
2007) and 0.5–1 mbar at 45°C (Hultquist et al. 2009). This value is less than the partial pressure of
H
2
present naturally in deep ground waters. In both Swedish and Finnish deep ground waters, the
concentration of dissolved H
2
is of the order of 10
–6
–10
–5
mol·dm
–3
(Auqué et al. 2006, King et al.
2001), equivalent to a partial pressure of 1.3–13 mbar. Under these conditions, corrosion of copper
by water is not thermodynamically favourable according to the data of Hultquist and co-workers.
Even if the ground water H
2
content is below the equilibrium partial pressure of H
2
, a significant
pressure of H
2
would quickly develop in the repository if the corrosion mechanism and corrosion
rate proposed by Hultquist et al. are correct. The repository is an almost completely closed system.
That, after all, is what it is designed for; to limit the escape of radionuclides from the repository
to the surface. To achieve this aim, highly compacted bentonite with a low hydraulic permeability
(<10
–13
m/s once fully saturated, SKB (2006c)) is used to seal the canisters in the boreholes or deposi-
tion tunnels. Dissolved and gaseous H
2
can pass through the bentonite by a number of mechanisms.
However, the rate of diffusion of dissolved H
2
is so low that, by analogy with the similarly imperme-
able Opalinus clay, the maximum rate of diffusive transport is equivalent to the rate at which H
2
would be produced by the corrosion in water at a rate of a few nm/yr (Nagra 2004). If H
2
is produced
any faster than this rate then a separate H
2
gas phase would form at the canister surface. Gaseous
H
2
could be transported by two-phase flow and, at higher pressure, by dilatancy of the pores in the
bentonite, but these processes operate at H
2
pressures of several MPa (i.e. 10’s of bar). Therefore,
if the rate of oxidation of copper by water exceeds a few nm/yr, a H
2
gas phase with a pressure of
several MPa would develop at the canister surface. This would clearly suppress corrosion if the H
2
equilibrium partial is of the order of 100’s of Pa (mbar). At this point, corrosion would only continue
if H
2
could escape the interfacial region, but evidence from the Opalinus clay studies indicates that
this would only occur by aqueous diffusion at the low gas pressures in question, with the flux of H
2
equivalent to a corrosion rate of a few nm/yr.
TR-10-69 43
4 Implications for canister lifetimes
It is clear from the discussion above that there are questions concerning both the scientific validity
of the observations reported by Hultquist and co-workers and, more importantly, their relevance to
the behaviour of copper canisters in a deep geological repository. In this section, these questions are
structured in the form of a decision tree (Figure 4-1). For the oxidation of copper by water to impact
the lifetimes of copper canisters, each of the questions must be answered “Yes.” A single “No”
answer means that the question of whether water can oxidise copper is not relevant to the use of
copper as a canister material in the KBS-3 design.
Based on the information presented by Hultquist and co-workers and by other researchers, as well as
the insights provided by the supplemental analyses, the answers for each of the four questions posed
in the decision tree are as follows:
Is there credible scientific evidence for the oxidation of copper by water?
The answer to this question is uncertain. There are certain observations and analyses that would
support the proposed mechanism, including:
• theobservationofH
2
using a solid electrolyte probe (Hultquist 1986), ion pump technique and/or
pressure increases (Hultquist et al. 2009, Szakálos et al. 2007),
• differencesinbehaviourofcopperexposedtowateringlassvesselssealedbyPdandbyPt
(Hultquist et al. 1989, 2008),
• thepossiblethermodynamicstabilityofsub-monolayerCuOH
ADS
species at potentials lower than
the Cu
2
O/H
2
O equilibrium line (Section 3.1).
However, there are also a number of observations and analyses that do not support the proposed
mechanism, including:
• thepoorcharacterisationofthestablesolidphase(H
x
CuO
y
) supposedly formed during the
corrosion of copper in water,
• thefactthatthecorrosionofcopperinwaterbecomesthermodynamicallyunfavourableinthe
presence of a monolayer of CuOH
ADS
(Section 3.1),
• thesuggestionthatonlyH
2
O, and not O
2
, is cathodically reduced in O
2
-containing water
(Gråsjö et al. 1995, Hultquist 1986, Hultquist et al. 2008, Seo et al. 1987, Szakálos et al. 2007),
• theuseofevidencefromgasphasestudiestoinferthebehaviourofO
2
and the composition of
corrosion products in aqueous solution,
• thesuggestionthatthereductionofH
2
O can cause the oxidation of Cu(I) to Cu(II)
(Hultquist et al. 1989), and
• theobservationthatthemeasuredcorrosionpotentialofcopperis150mVmorepositivethan
the equilibrium potential for the H
2
evolution reaction at 1 atm pressure (Seo et al. 1987).
If there is credible evidence for the oxidation of copper by water, has the effect been repro-
duced by other researchers?
The answer to this question is “No.” Despite three published attempts at reproducing various aspects
of the experiments by Hultquist and co-workers, no other researcher has managed to find any
evidence that water can oxidise copper. These other studies include:
• anattemptbySimpsonandSchenk(1987)tomeasureH
2
due to the corrosion of copper in
O
2
-free Cl
–
solutions (Section 2.1.2),
• anattemptbyEriksenetal.(1988,1989)tomeasureH
2
due to the corrosion of copper in O
2
-free
water (Section 2.1.4), and
• anattemptbyMöller(1995)toreproducethedifferencesobservedbyHultquistetal.(1989)
between the appearance of copper exposed to water in Pd-sealed and Pt-sealed glass vessels
(Section 2.1.6).

44 TR-10-69
Figure 4-1.
Decision tree approach to the question of whether the oxidation of copper by water is an
important process for the KBS-3 repository design.
Is there
credible scientific evidence
for the oxidation of copper by
water?
No
The oxidation of copper by water is not an important process for the KBS-3 repository design
If there is
scientific evidence for
the oxidation of copper by
water, has the effect been
reproduced by other
researchers?
If water is
an oxidant for copper, is the
mechanism relevant for
repository conditions?
Yes
Yes
If oxidation of
copper by water is possible
in the repository, will canister
lifetimes be adversely
affected?
No
Yes
Evidence for H
2
formation
and differences between Pd-
sealed and Pt-sealed tests
support proposed
mechanism, BUT poorly
characterised corrosion
product H
x
CuO
y
, proposed
preferential reduction of H
2
O
over O
2
, E
CORR
measurements, and other
factors count against it
No
No
Three separate groups
have failed to
reproduce H
2
production or
differences between
Pd-sealed and Pt-
sealed tests
Unproven mechanism
in water even more
unlikely in Cl
-
pore
water solution.
Background p
H2
or
development of H
2
gas
phase will suppress
copper oxidation.
Effect of H
2
O reduction
due to sulphide already
taken into account.
Maximum rate due to
oxidation by water
determined by slow
diffusion of dissolved
H
2
(~nm/yr).
Yes
TR-10-69 45
Furthermore, Bojinov and Mäkelä (2003) and Bojinov et al. (2004) could not detect H
2
in their
single attempt from copper corrosion experiments in anoxic concentrated Cl
–
solution. Furthermore,
in situ measurement of the corrosion rate and corrosion potential suggested that corrosion ceased
once the potential reached the thermodynamically predicted immunity zone (Section 3.2).
This inability of other researchers to reproduce the results of Hultquist and co-workers is the most
troubling aspect of this issue. One of the fundamental principles of experimental scientific work
is that the observations should be repeatable and reproducible. Hultquist and co-workers have not
provided an adequate explanation for this failure on the part of others to reproduce their results.
This failure is particularly concerning in the case of Simpson and Schenk who had developed prior
expertise in the use of H
2
evolution measurements to study the corrosion of C-steel.
If water is an oxidant for copper, is the proposed mechanism relevant to conditions in a deep
geological repository?
The answer to this question is “No.” Notwithstanding the absence of a proven mechanism by which
water oxidises copper in the laboratory, it is clear that the proposed mechanism is not relevant to the
conditions in the repository. Even under laboratory conditions, H
2
production has only been claimed
in bulk water. The canister in the repository is surrounded by highly compacted bentonite with saline
pore fluids and saline ground water. The Cl
–
ions in the pore solution and ground water determine the
anodic dissolution behaviour of copper and determine the nature of the corrosion products formed
(Sections 3.1–3.4). No evidence for H
2
evolution has been observed in two studies of the corrosion
of copper in anoxic Cl
–
solutions (Bojinov and Mäkelä 2003, Bojinov et al. 2004, Simpson and
Schenk 1987).
Perhaps the most important reason that the proposed mechanism is not important under repository
conditions is because the partial pressure of H
2
in deep Swedish and Finnish ground waters is higher
than the proposed equilibrium H
2
partial pressure at 73°C. Furthermore, because of the low hydraulic
conductivity of highly compacted bentonite, a H
2
gas phase would quickly develop around the
container if corrosion in water did occur, which would suppress any further corrosion to a rate of a
few nm/yr, equivalent to the diffusive flux of dissolved H
2
away from the canister surface.
If the oxidation of copper by water in the repository is possible, will it adversely affect the
canister lifetime?
The answer to this question is “No.” It should be remembered that corrosion of the canister sup-
ported by the reduction of water is already included in the safety analysis for the KBS-3 concept
(SKB 2006a, b). Sulphide present in the deposition hole or that diffusing through the bentonite from
the ground water is assumed to reach the canister surface causing corrosion and, because of the
stability of the Cu
2
S that is formed, the evolution of H
2
from the reduction of water. Because of the
limited amount of sulphide available in the repository and because of the limited rate of supply to
the canister surface because of the highly compacted bentonite, this process is predicted to result in
<5 mm of corrosion in a period of 100,000 years (SKB 2006a, b).
Any additional corrosion that would occur if the proposed oxidation of copper water were to take
place would be minor in comparison. If the H
2
partial pressure in the ground water at repository
depth exceeds the equilibrium partial pressure for the oxidation of copper, then no corrosion via this
process will occur. If the ground water H
2
partial pressure is insufficient to suppress corrosion by
this mechanism, then corrosion would proceed for a short period until such time that a H
2
gas phase
developed at the canister surface. Based on analyses of the H
2
transport capacity of Opalinus clay
(which has a similar hydraulic conductivity to highly compacted bentonite, Nagra (2004)), such a
gas phase would be expected to develop if the corrosion rate exceeds a few nm/yr. At this corrosion
rate, the rate of H
2
generation exceeds the rate at which dissolved H
2
could be removed from the
canister surface by diffusion through the bentonite. In this almost-completely sealed system, corro-
sion would continue at the rate at which H
2
could be transported away from the interface, i.e. a rate
of a few nm/yr corresponding to the rate of diffusion of dissolved H
2
.
TR-10-69 47
5 Summary and conclusions
A critical review of the literature in support of and against the suggestion that water oxidises copper
has been carried out. The important findings and conclusions from the review include:
• Hultquistandco-workershaveprovidedevidencefortheevolutionofH
2
during the corrosion
of copper in pure water based on: (i) the difference in corrosion rate and visual appearance of
samples in Pd- and Pt-sealed vessels, (ii) ion-pump measurements of the flux of H
2
through a
Pd membrane, (iii) pressure changes due to the consumption of O
2
and the production of H
2
.
• Gråsjöetal.(1995)didnotobserveH
2
production from the corrosion of copper in water,
although the experiment may not have been allowed to run long enough for anoxic conditions
to be established.
• NeitherSimpsonandSchenk(1987)norEriksenetal.(1988,1989)observedH
2
production
from the corrosion of copper in aqueous chloride solutions or pure water, respectively, using
gas chromatography.
• Möller(1995)didnotseeanyvisualdifferenceintheappearanceofCustripsinPd-and
Pt-sealed glass vessels and corrosion appears to have ceased half-way through the 2-year
experiment.
• ThepotentialofCuwireduringsupposedH
2
generation is 150 mV more positive than the
equilibrium potential for the H
2
O/H
2
rection for a H
2
pressure of 1 atm (Seo et al. 1987).
• TheproductionofH
2
has been associated with the formation of a previously unknown phase
H
x
CuO
y
(Hultquist et al. 2008, 2009, Szakálos et al. 2007).
• TheequilibriumH
2
pressure for the Cu/ H
x
CuO
y
system is believed to be ~1 mbar (101 Pa) at
73°C (Szakálos et al. 2007).
• Iftheion-pumpcurrentdescribedintheworkofSzakálosetal.(2007)isameasureofthefluxof
H
2
as the authors claim, then their results indicate that the rate of corrosion increases monotoni-
cally upon exposure to O
2
-free water.
• Hultquistandco-workerspointtothepresenceofHincorrosionproductsoncopperasevidence
for the reduction of H
2
O, but passive films, including those on copper, are typically hydrated to
some degree.
• ThecorrosionofcopperinwaterwillcontinueprovidedH
2
is removed from the interface and the
pressure remains below the equilibrium pressure (Hultquist 1986, Hultquist et al. 1989, Szakálos
et al. 2007).
• Oxygendoesnotsupportthecorrosionofcopperinwaterbutis,instead,consumedbyreaction
with H atoms produced by the reduction of H
2
O (Gråsjö et al. 1995, Hultquist 1986, Hultquist
et al. 2008, Seo et al. 1987, Szakálos et al. 2007).
• ThereductionofH
2
O is capable of supporting the oxidation of copper to the Cu(I) and Cu(II)
oxidation states (Hultquist et al. 1989).
• BasedontheanalysisbytheSSMBRITEgroupofthepaperbySzakálosetal.(2007),thecorro-
sion rate of copper in the "pressure-gauge" experiment is approximately 40 times lower than that
in the ion-pump experiment despite being done at a higher temperature.
• Quantummechanicalcalculationsindicatethattherearenostable(withrespecttoCu
2
O)
compounds that would correspond to the proposed H
x
CuO
y
species proposed by Hultquist and
co-workers (Korzhavyi and Johansson 2010).
• Bojinovetal.(2010)suggestthatH
2
could be formed without the oxidation of copper due to
reactions between an adsorbed CuOH
ads
species and H
2
O. The CuOH
ads
species is formed from
the reduction of dissolved Cu
2+
that results from the dissolution of a pre-formed Cu
2
O layer or
from the consumption of residual O
2
.
48 TR-10-69
• Hultquistandco-workershavereportedH
2
generation from initially aerated experiments (e.g.
Hultquist (1986)) and from experiments in which the water used had been extensively deaerated
(e.g. Hultquist et al. (2009)). Thus, pre-oxidation of the surface does not seem to affect H
2
generation from copper.
• NeitherHultquistandco-workersnorotherresearchershavereportedthegenerationofH
2
when
copper is exposed to solutions other than pure water, e.g. saline solutions representative of
bentonite pore water or deep ground waters.
A decision-tree approach has been used to summarise the findings of the critical review and to
address the relevance of these findings for the corrosion behaviour of the canister. It is found that:
• thescientificevidenceinsupportofthesuggestionthatwateroxidisescopperisnotconclusive
and there are many aspects which are unclear and contradictory,
• despiteanumberofattempts,nootherresearchershavebeenabletoreproducetheobservations
of Hultquist and co-workers,
• evenifcorrect,themechanismisnotimportantforcoppercanistersinarepository,bothbecause
of differences in the environmental conditions and because, even if corrosion did occur by this
mechanism, it would quickly stop, and
• thereisnoadverseimpactonthelifetimeofcoppercanistersduetothisproposed,butunproven,
mechanism.
TR-10-69 49
References
SKB’s (Svensk Kärnbränslehantering AB) publications can be found at www.skb.se/publications.
Apted M J, Bennett D G, Saario T, 2009. A review of evidence for corrosion of copper by water.
SSM Report 2009:30, Strålsäkerhetsmyndigheten (Swedish Radiation Safety Authority).
Auqué L F, Gimeno M J, Gómez J B, Puigdomenech I, Smellie J, Tullborg E-L, 2006.
Groundwater chemistry around a repository for spent nuclear fuel over a glacial cycle. Evaluation
for SR-Can. SKB TR-06-31, Svensk Kärnbränslehantering AB.
Battino R, Rettich T R, Tominaga T, 1983. The solubility of oxygen and ozone in liquids. Journal
of Physical and Chemical Reference Data, 12, pp 163–178.
Bojinov M, Mäkelä K, 2003. Corrosion of copper in anoxic 1M NaCl solution. Posiva Working
Report 2003-45, Posiva Oy, Finland.
Bojinov M, Laitinen T, Mäkelä K, Snellman M, Werme L, 2004. Corrosion of copper in 1 M
NaCl under strictly anoxic conditions. In: Oversby V M, Werme L O (eds). Scientific basis for nuclear
waste management XXVII: symposium held in Kalmar, Sweden, 15–19 June 2003. Warrendale, PA:
Materials Research Society. (Materials Research Society Symposium Proceedings 807).
Bojinov M, Betova I, Lilja C, 2010. A mechanism of interaction of copper with a deoxygenated
neutral aqueous solution. Corrosion Science, 52, pp 2917–2927.
Burke L D, Murphy M A, 2001. Multilayer hydrous oxide growth on copper in base and its
correlation with previously reported electrocatalytic data for this electrode system. Journal of Solid
State Electrochemistry, 5, pp 43–49.
Burke L D, Ahern M J G, Ryan T G, 1990. An investigation of the anodic behavior of copper
and its anodically produced oxides in aqueous solutions of high pH. Journal of the Electrochemical
Society, 137, pp 553–561.
Burstein G T, Newman R C, 1981. Reactions of scratched copper electrodes in aqueous solutions.
Journal of the Electrochemical Society, 128, pp 2270–2276.
Butler I R, Schoonen M A A, Rickard D T, 1994. Removal of dissolved oxygen from water: a
comparison of four common techniques. Talanta 41, pp 211–215.
Corbett R A, Elliot P, 2000. Ant-nest corrosion – digging the tunnels. In: Corrosion/2000, NACE
International, Houston, Texas, Paper 646.
Elsner C I, Salvarezza R C, Arvia A J, 1988. The influence of halide ions at submonolayer levels
on the formation of oxide layer and electrodissolution of copper in neutral solutions. Electrochimica
Acta, 33, pp 1735–1741.
Eriksen T E, Ndalamba P, Grenthe I, 1988. On the corrosion of copper in pure water. SKB
TR 88-17, Svensk Kärnbränslehantering AB.
Eriksen T E, Ndalamba P, Grenthe I, 1989. On the corrosion of copper in pure water. Corrosion
Science, 29, pp 1241–1250.
Figueroa M G, Salvarezza R C, Arvia A J, 1986. The influence of temperature on the pitting
corrosion of copper. Electrochimica Acta, 31, pp 665–669.
Fitzsimons N P, Jones W, Herley P J, 1995. Studies of copper hydride. Part 1 – synthesis and solid
state stability. Journal of the Chemical Society, Faraday Transactions, 91, pp 713–718.
Gråsjö L, Hultquist G, Lu Q, Seo M, 1995. In situ measurements of oxygen consumption and hydrogen
evolution in corrosion of some materials in liquid water. Materials Science Forum, 185–188, pp 703–712.
Gu B, Luo J and Mao X, 1999. Hydrogen-facilitated anodic dissolution-type stress corrosion
cracking of pipeline steels in near-neutral pH solution. Corrosion, 55, pp 96–106.
Hänninen H, Forsling W, 2007. A few comments on the published study “Corrosion of Copper by
Water” by P. Szakálos, G. Hultquist, and G. Wikmark. [Online]. Available at: http://www.mkg.se/
pdf/Karnavfallsradet_PM_Hanninen_Forsling_Kommentarer_om_kopparkorrosion.pdf.
50 TR-10-69
Hultquist G, 1986. Hydrogen evolution in corrosion of copper in pure water. Corrosion Science, 26,
pp 173–177.
Hultquist G, Chuah G K, Tan K L, 1989. Comments on hydrogen evolution from the corrosion of
pure copper. Corrosion Science, 29, pp 1371–1377.
Hultquist G, Gråsjö L, Lu Q, 1993. Consumption and release of gas components during the
reaction of iron at 300°C in a H
2
16
O/H
2
18
O/O
2
gas mixture. Corrosion Science, 34, pp 1035–1038.
Hultquist G, Gråsjö L, Lu Q, Åkermark T, 1994. The analysis of gas consumption in the reaction
of Fe and Cu in H
2
16
O/H
2
18
O/O
2
gas mixtures. Corrosion Science, 36, pp 1459–1471.
Hultquist G, Szakálos P, Graham M J, Sproule G I, Wikmark G, 2008. Detection of hydrogen in
corrosion of copper in pure water. In: Proceedings of the 17th International Corrosion Congress, Las
Vegas, Nevada, September 2008. Houston, TX: NACE International, Paper 3884.
Hultquist G, Szakálos P, Graham M J, Belonoshko A B, Sproule G I, Gråsjö L,
Dorogokupets P, Danilov B, AAstrup T, Wikmark G, Chuah G-K, Eriksson J-C, Rosengren A,
2009. Water corrodes copper. Catal. Lett. 132, 311–316.
Johansson L-G, 2008. Comment on “corrosion of copper by water” [Electrochem. Solid-State Lett.,
10, C63, (2007)]. Electrochemical Solid-State Letters, 11, pp S1–S1.
Kear G, Barker B D, Walsh F C, 2004. Electrochemical corrosion of unalloyed copper in chloride
media – a critical review. Corrosion Science, 46, pp 109–135.
King F, 2002. Corrosion of copper in alkaline chloride environments. SKB TR-02-25, Svensk
Kärnbränslehantering AB.
King F, Kolár M, 2000. The copper container corrosion model used in AECL’s second case study.
Report 06819-REP-01200-10041-R00, Ontario Power Generation, Nuclear Waste Management
Division, Canada.
King F, Quinn M J, Litke C D, 1995a. Oxygen reduction on copper in neutral NaCl solution.
Journal of Electroanalytical Chemistry, 385, pp 45–55.
King F, Litke C D, Tang Y, 1995b. Effect of interfacial pH on the reduction of oxygen on copper
in neutral NaClO4 solution. Journal of Electroanalytical Chemistry, 384, pp 105–113.
King F, Litke C D, Quinn M J, LeNeveu D M, 1995c. The measurement and prediction of the
corrosion potential of copper in chloride solutions as a function of oxygen concentration and mass-
transfer coefficient. Corrosion Science, 37, pp 833–851.
King F, Ryan S R, Litke C D, 1997. The corrosion of copper in compacted clay. AECL-11831,
COG-97-319-I, Atomic Energy of Canada Limited.
King F, Ahonen L, Taxén C, Vuorinen U, Werme L, 2001. Copper corrosion under expected
conditions in a deep geologic repository. SKB TR-01-23, Svensk Kärnbränslehantering AB.
Korzhavyi P A, Johansson B, 2010. Thermodynamic properties of copper compounds with oxygen
and hydrogen from first principles. SKB TR-10-30, Svensk Kärnbränslehantering AB.
Maurice V, Strehblow H-H, Marcus P, 2000. In situ STM study of the initial stages of oxidation
of Cu(III) in aqueous solution. Surface Science, 458, pp 185–194.
Möller K, 1995. Kopparkorrosion i rent syrefritt vatten. SKI Rapport 95:72, Statens
kärnkraftinspektion (Swedish Nuclear Power Inspectorate). (In Swedish.)
Moreau A, 1981. Etude du mecanisme d’oxydo-reduction du cuivre dans les solutions chlorurees
acides-I. Systeme Cu-. Electrochimica Acta, 26, pp 497–504.
Nagra, 2004. Effects of post-disposal gas generation in a repository for spent fuel, high-level waste
and long-lived intermediate level waste sited in Opalinus Clay. Nagra Technical Report NTB 04-06,
National Cooperative for the Disposal of Radioactive Waste, Switzerland.
Panagopoulos C N, Zacharopoulos N, 1994. Cathodic hydrogen charging and mechanical
properties of copper. Journal of Material Science, 29, pp 3843–3846.
TR-10-69 51
Pourbaix M, 1974. Atlas of electrochemical equilibria in aqueous solutions. 2nd ed. Houston,
TX: NACE.
Protopopoff E, Marcus P, 2005. Potential-pH diagrams for hydroxyl and hydrogen adsorbed on a
copper surface. Electrochimica Acta, 51, pp 408–417.
Puigdomenech I, Taxén C, 2000. Thermodynamic data for copper. Implications for the corrosion
of copper under repository conditions. SKB TR-00-13, Svensk Kärnbränslehantering AB.
San Marchi C, 2006. Technical reference on hydrogen compatibility of materials. Copper
alloys: pure copper (code 4001). [Online]. Available at: http://www.sandia.gov/matlsTechRef/
chapters/4001TechRef_Cu.pdf.
Sandia, 2000. Sandia researchers perform first-ever multiple copper corrosion experiments on
single. Sandia National Laboratories, Science Daily, [Online] 7 August. Available at:
http://www.sciencedaily.com/releases/2000/08/000807070959.htm.
Sandström M, Jalilehvand F, Persson I, Gelius U, Frank P, Hall-Roth I, 2002. Deterioration of the
seventeenth-century warship Vasa by internal formation of sulphuric acid. Nature, 415, pp 893–897.
Seo M, Hultquist G, Gråsjö L, Sato N, 1987. Hydrogen evolution kinetics in corrosion of copper,
iron and zinc in water. In: Proceedings of the 10th International Congress on Metallic Corrosion,
Madras, India, November 1987. Trans Tech Publications, Switzerland, paper 2.31, pp 481–488.
Sharma V K, Millero F J, 1988. The oxidation of Cu(I) in electrolyte solutions. Journal of Solution
Chemistry, 17, pp 581–599.
Simpson J P, Schenk R, 1987. Hydrogen evolution from corrosion of pure copper. Corrosion
Science, 27, pp 1365–1370.
SKB, 2006a. Long-term safety for KBS-3 repositories at Forsmark and Laxemar – a first evaluation.
Main report of the SR-Can project. SKB TR-06-09, Svensk Kärnbränslehantering AB.
SKB, 2006b. Fuel and canister process report for the safety assessment SR-Can. SKB TR-06-22,
Svensk Kärnbränslehantering AB.
SKB, 2006c. Buffer and backfill process report for the safety assessment SR-Can. SKB TR-06-18,
Svensk Kärnbränslehantering AB.
Spitzer A, Lüth H, 1985. An XPS study of the water adsorption on Cu(110). Surface Science, 160,
pp 353–361.
Szakálos P, Hultquist G, 2008. Correspondence with Drs. H. Hänninen and W. Forsling, Swedish
National Council for Nuclear Waste. [Online]. Available at: http://www.mkg.se/pdf/Szakalos_m_fl_
svar_till_Karnavfallsradet_080229.pdf.
Szakálos P, Hultquist G, Wikmark G, 2007. Corrosion of copper by water. Electrochemical Solid-
State Letters, 10, pp C63–C67.
Szakálos P, Hultquist G, Wikmark G, 2008. Response to the comment on “corrosion of copper by
water” [Electrochem. Solid-State Lett., 10, C63, (2007)]. Electrochemical Solid-State Letters, 11,
pp S2–S2.
Vazquez M V, de Sanchez S R, Calvo E J, Schiffrin D J, 1994a. The electrochemical reduction
of oxygen on polycrystalline copper in borax buffer. Journal of Electroanalytical Chemistry, 374,
pp 189–197.
Vazquez M V, de Sanchez S R, Calvo E J, Schiffrin D J, 1994b. The electrochemical reduction of
hydrogen peroxide on polycrystalline copper in borax buffer. Journal of Electroanalytical Chemistry,
374, pp 179–187.
Wagman D D, Evans W H, Parker V B, Schumm R H, Halow I, Bailey S M, Churney K L,
Nuttall R L, 1982. The NBS tables of chemical thermodynamic properties. Selected values for
inorganic and C
1
and C
2
organic substances in SI units. Journal of Physical and Chemical Reference
Data, 11, Supplement 2.
Yang Q, Qiao L J, Chiovelli S, Luo J L, 1998. Effects of hydrogen on pitting susceptibility of
Type 310 stainless steel. Corrosion, 54, pp 628–633.
